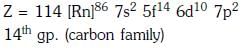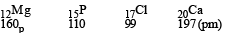31 Years NEET Previous Year Questions: Classification of Elements & Periodicity
MCQ Practice Test & Solutions: 31 Years NEET Previous Year Questions: Classification of Elements & Periodicity in Properties (30 Questions)
You can prepare effectively for NEET Chemistry Class 11 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "31 Years NEET Previous Year Questions: Classification of Elements & Periodicity in Properties". These 30 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 30 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
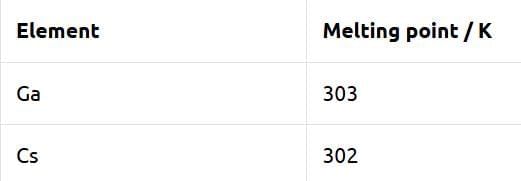
Which of the following statements are true? [NEET 2025]

Choose the correct answer from the options given below:

Detailed Solution: Question 1
Which among the following electronic configurations belong to main group elements? [NEET 2025]
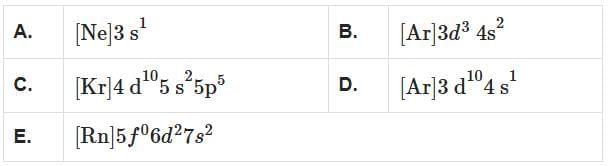
Choose the correct answer from the option given below:

Detailed Solution: Question 2
Arrange the following elements in increasing order of electronegativity: [NEET 2024]
N, O, F, C, Si
Choose the correct answer from the options given below :
N, O, F, C, Si
Choose the correct answer from the options given below :
Detailed Solution: Question 3
Arrange the following elements in increasing order of first ionization enthalpy: [NEET 2024]
Li, Be, B, C, N
Choose the correct answer from the options given below:
Li, Be, B, C, N
Choose the correct answer from the options given below:
Detailed Solution: Question 4
Identify the incorrect statement: [NEET 2024]
Detailed Solution: Question 5
Match List-I with List-II: [NEET 2024]
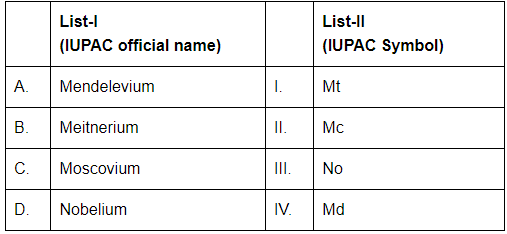
Choose the correct answer from the options given below:

Detailed Solution: Question 6
The correct decreasing order of atomic radii (pm) of Li, Be, B, and C is: [NEET 2024]
Detailed Solution: Question 7
Match List-I with List-II [NEET 2024]
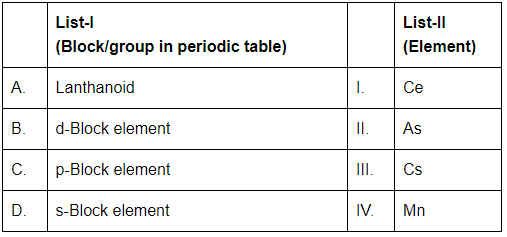
Choose the correct answer from the options given below:

Detailed Solution: Question 8
The element expected to form the largest ion to achieve the nearest noble gas configuration is: [NEET 2023]
Detailed Solution: Question 9
Which one of the following represents all isoelectronic species? [NEET 2023]
Detailed Solution: Question 10
Which of the following pairs is correctly matched? [NEET 2023]
Detailed Solution: Question 11
The correct sequence given below contains neutral, acidic, basic, and amphoteric oxide each, respectively is: [NEET 2023]
Detailed Solution: Question 12
The IUPAC name of an element with atomic number 119 is [NEET 2022 Phase 1]
Detailed Solution: Question 13
Gadolinium has a low value of third ionisation enthalpy because of [NEET 2022 Phase 1]
Detailed Solution: Question 14
The correct order of first ionization enthalpy for the given four elements is : [NEET 2022 Phase 2]
Detailed Solution: Question 15
Decrease in size from left to right in actinoid series is greater and gradual than that in lanthanoid series due to: [NEET 2022 Phase 2]
Detailed Solution: Question 16
Fluorine is a stronger oxidising agent than chlorine because : [NEET 2022 Phase 2]
(a) F-F bond has a low enthalpy of dissociation.
(b) Fluoride ion (F−) has high hydration enthalpy.
(c) Electron gain enthalpy of fluorine is less negative than chlorine.
(d) Fluorine has a very small size.
Choose the most appropriate answer from the options given :
(a) F-F bond has a low enthalpy of dissociation.
(b) Fluoride ion (F−) has high hydration enthalpy.
(c) Electron gain enthalpy of fluorine is less negative than chlorine.
(d) Fluorine has a very small size.
Choose the most appropriate answer from the options given :
Detailed Solution: Question 17
Identify the incorrect match. [NEET 2020]
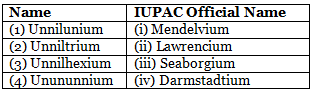

Detailed Solution: Question 18
Match the following: [NEET 2020]
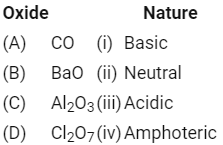
Which of the following is correct option?

Which of the following is correct option?
Detailed Solution: Question 19
For the second period elements the correct increasing order of first ionisation enthalpy is: [NEET 2019]
Detailed Solution: Question 20
The correct order of atomic radii in group 13 elements is [NEET 2018]
Detailed Solution: Question 21
Which of the following oxides is most acidic in nature? [NEET 2018]
Detailed Solution: Question 22
The element Z = 114 has been discovered recently. It will belong to which of the following family/groupand electronic configuration ? [NEET 2017]
Detailed Solution: Question 23
The species Ar, K+ and Ca2+ contain the same number of electrons. In which order do their radii increase ? [AIPMT 2015 Cancelled Paper]
Detailed Solution: Question 24
Which of the following orders of ionic radii is correctly represented? [NEET 2014]
Detailed Solution: Question 25
Which one of the following arrangements represents the correct order of least negative to most negative electron gain enthalpy for C, Ca, Al, F and O? [NEET Kar. 2013]
Detailed Solution: Question 26
Identify the wrong statement in the following: [2012]
Detailed Solution: Question 27
What is the value of electron gain enthalpy of Na+ if IE1 of Na = 5.1 eV ? [2011M]
Detailed Solution: Question 28
Among the elements Ca, Mg, P and Cl, the order of increasing atomic radii is : [2010]
Detailed Solution: Question 29
Which of the following represents the correct order of increasing electron gain enthalpy with negative sign for the elements O, S, F and Cl ? [2010]
Detailed Solution: Question 30
96 videos|243 docs|71 tests |