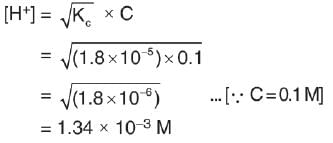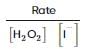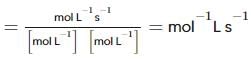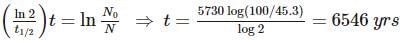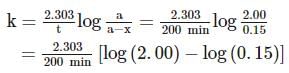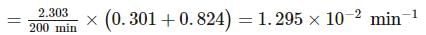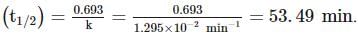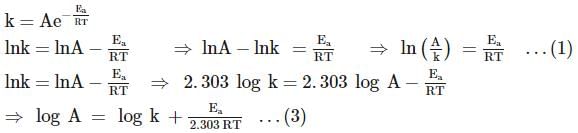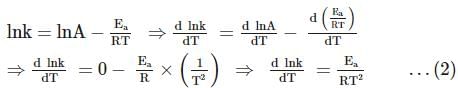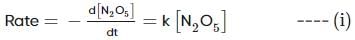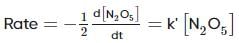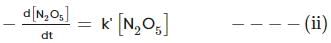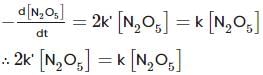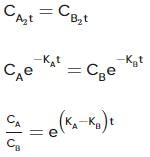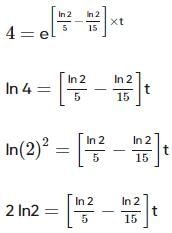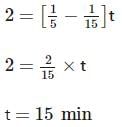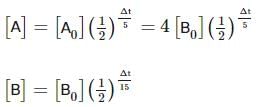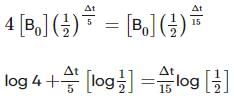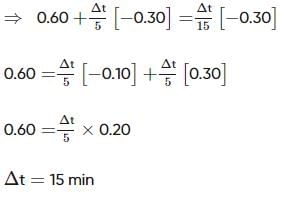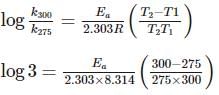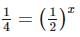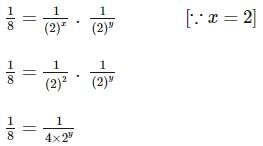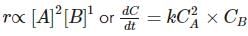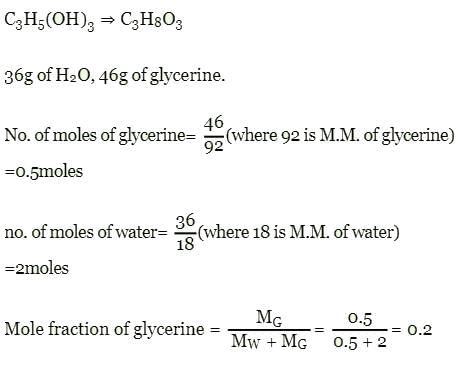BITSAT Chemistry Test - 1 Free Online Test 2026
MCQ Practice Test & Solutions: BITSAT Chemistry Test - 1 (30 Questions)
You can prepare effectively for JEE BITSAT Mock Tests Series & Past Year Papers 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "BITSAT Chemistry Test - 1". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 40 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
Detailed Solution: Question 2
Detailed Solution: Question 3
When primary amine is heated with CS2 in presence of excess of mercuric chloride, it produce isothiocyanate. This reaction is known as
Detailed Solution: Question 4
Detailed Solution: Question 5
Total number of electrons in all the p-orbitals of bromine will be
Detailed Solution: Question 6
Detailed Solution: Question 7
Ionization constant of acetic acid is 1.8 * 10-5. The concentration of H+ ions in 0.1 M solution is
Detailed Solution: Question 8
Detailed Solution: Question 9
On heating one end of a piece of metal, tlhe other end becomes hot because of
Detailed Solution: Question 10
From the rate expression for the following reaction, determine its order of reaction and the dimensions of the rate constant.
H2O2(aq)+3I−(aq)+2H+⟶2H2O(l)+I−3;Rate=k[H2O2][I−]
H2O2(aq)+3I−(aq)+2H+⟶2H2O(l)+I−3;Rate=k[H2O2][I−]
Detailed Solution: Question 11
An archaeologist unearths a bone sample and wants to know the age of the bone. Her chemist friend determines the 45.3% of the initial amount of carbon-14 is present in the bone sample. If the half-life of carbon-14 is 5730 yrs, calculate the age (in years) of the bone.
Detailed Solution: Question 12
A substance 'A' decomposes by a first order reaction starting initially with [A]=2.00 M and after 200 min, [A] becomes 0.15 M . For this reaction, t1/2 is
Detailed Solution: Question 13
Detailed Solution: Question 14
For the first-order decomposition reaction of N2O5 , it is observed that,
(i) N2O5(g)→2NO2(g)+1/2O2(g);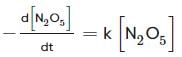
(ii) 2N2O5(g)→4NO2(g)+O2(g);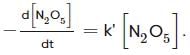
Which of the following is true?
(i) N2O5(g)→2NO2(g)+1/2O2(g);
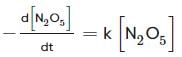
(ii) 2N2O5(g)→4NO2(g)+O2(g);
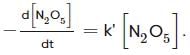
Detailed Solution: Question 15
What should be the age of the fossil for meaningful determination of its age?
Detailed Solution: Question 16
Two substances A (t1/2=5 min) and B (t1/2=15 min) are taken in such a way that initially [A]=4[B] . The time after which both the concentration will be equal is : (Assume that reaction is first order)
Detailed Solution: Question 17
In the start of summer, a given sample of milk turns sour at room temperature (27oC) in 48 hour . In a refrigerator at 2oC , milk can be stored three times before it sours.
For souring of milk, At 2oC , the reaction is three times slower than at 27oC . The activation energy of the souring of milk is (in kJ mol−1) .
For souring of milk, At 2oC , the reaction is three times slower than at 27oC . The activation energy of the souring of milk is (in kJ mol−1) .
Detailed Solution: Question 18
A + B → Product
If concentration of A is doubled, rate increases 4 times. If concentration of A and B are doubled, rate increases 8 times. The differential rate equation of the reaction will be
If concentration of A is doubled, rate increases 4 times. If concentration of A and B are doubled, rate increases 8 times. The differential rate equation of the reaction will be
Detailed Solution: Question 19
Detailed Solution: Question 20
According to quantum mechanics ψ2(r) the wave function squared gives
Detailed Solution: Question 21
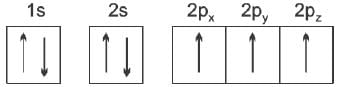
Spin quantum number with two spin states of the electron represented by two arrows, ↑ (spin up) and ↓ (spin down) was introduced to account for
Detailed Solution: Question 22
Detailed Solution: Question 23
The reagents to bring about the change from but – 2 – ene to ethanal
Detailed Solution: Question 24
Detailed Solution: Question 25
Detailed Solution: Question 26
Which of the following reagents can be used to convert a carboxylic acid directly into its corresponding acid chloride derivative?
Detailed Solution: Question 27
Commercial concentrated nitric acid is 15.6 M. To prepare 10 L of 6.0 M nitric acid from it,
Detailed Solution: Question 28
Mole fraction of C3H5(OH)3 in a solution of 36 gm of water and 46 gm of glycerine is
Detailed Solution: Question 29
AgNO3 sample is 85% by mass. To prepare 125 mL of 0.05 M AgNO3 solution, AgNO3 sample required is
Detailed Solution: Question 30
2 videos|18 docs|70 tests |