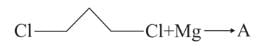
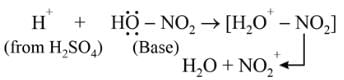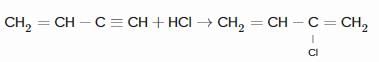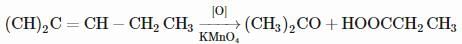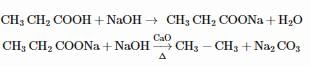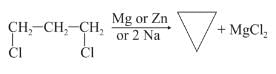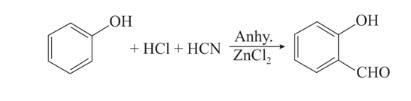BITSAT Chemistry Test - 4 Free Online Test 2026
MCQ Practice Test & Solutions: BITSAT Chemistry Test - 4 (30 Questions)
You can prepare effectively for JEE BITSAT Mock Tests Series & Past Year Papers 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "BITSAT Chemistry Test - 4". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 40 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following group is sharp ortho and para directive?
Detailed Solution: Question 1
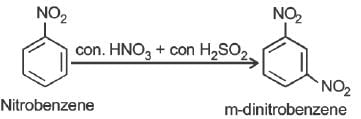
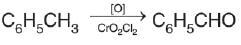
Nitrobenzene can be prepared from benzene by using a mixture of concentrated HNO3 and concentrated. H2SO4. In the mixture, nitric acid acts as a/an:
Detailed Solution: Question 2
Detailed Solution: Question 3
On vigorous oxidation by alkaline permanganate solution, (CH3)2C=CH−CH2CH3 gives:
Detailed Solution: Question 4
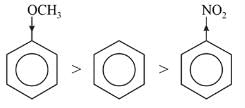
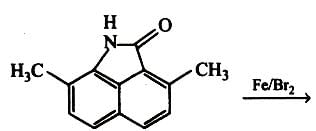
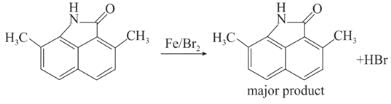
Among the following compounds (I-III) the correct order of reaction with electrophilic reagent is
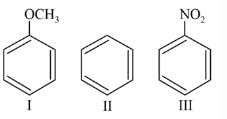

Detailed Solution: Question 5
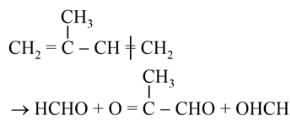
The products obtained on ozonolysis of the given alkene, followed by reaction with Zn/H2O are:
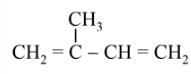
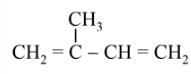
Detailed Solution: Question 6
Detailed Solution: Question 7
The number of geometrical isomers of CH3CH=CH−CH=CH−CH=CHCl is
Detailed Solution: Question 8
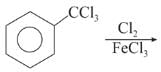
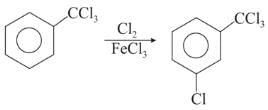
Chlorination of benzene is not possible in the following reaction.
Detailed Solution: Question 9
Detailed Solution: Question 10
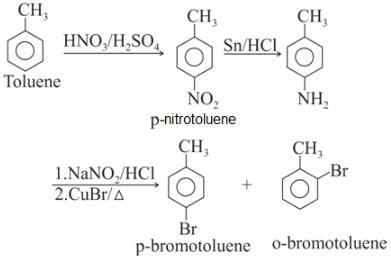
Toluene is nitrated and the resulting product is reduced with tin and hydrochloric acid. The product so obtained is mixed with NaNO2/HCl and CuBr and then heated, the product so formed contains.
Detailed Solution: Question 11
Detailed Solution: Question 12
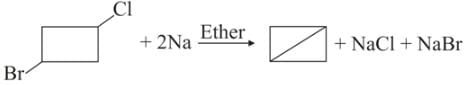
What would be the product formed when 1-bromo-3-chlorocyclobutane reacts with two equivalents of metallic sodium in ether?
Detailed Solution: Question 13
Detailed Solution: Question 14
Which of the following undergoes nucleophilic substitution exclusively by an SN1 mechanism
Detailed Solution: Question 15
Detailed Solution: Question 16
Detailed Solution: Question 17
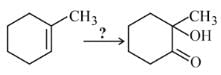
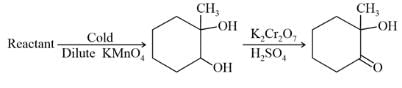
In the following sequence of reactions, 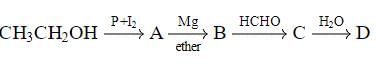
, then compound D is:
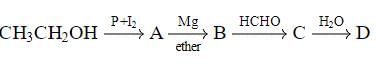
Detailed Solution: Question 18
Detailed Solution: Question 19
Which of the following compounds will form the precipitate with aq. AgNO3 solution most readily?
Detailed Solution: Question 20
Detailed Solution: Question 21
Detailed Solution: Question 22
The specific rate constant of a first order reaction depends on
Detailed Solution: Question 23
The law which explains the law of conservation of mass is
Detailed Solution: Question 24
Detailed Solution: Question 25
Two molecules of an ideal gas expand spontaneously into a vacuum. The work done is
Detailed Solution: Question 26
Detailed Solution: Question 27
Detailed Solution: Question 28
At 25°C the pH value of a solution is 6, the solution is
Detailed Solution: Question 29
Detailed Solution: Question 30
2 videos|18 docs|70 tests |