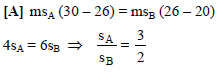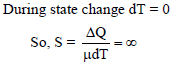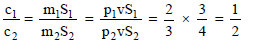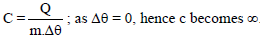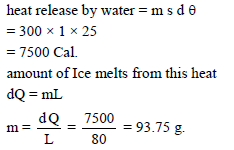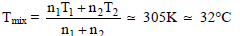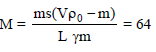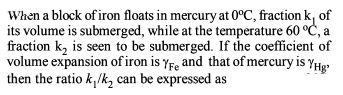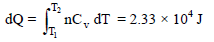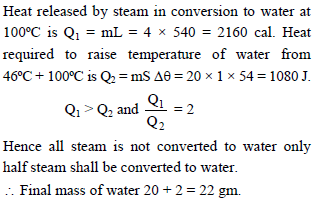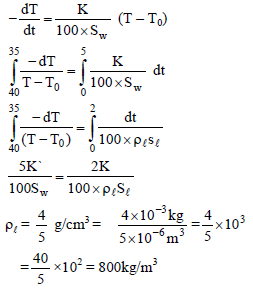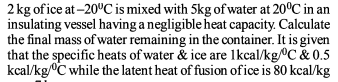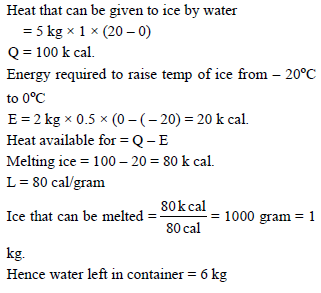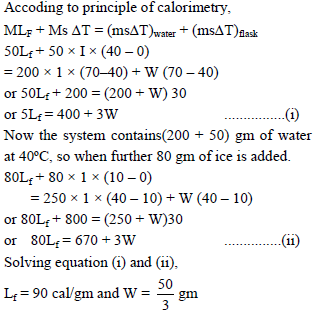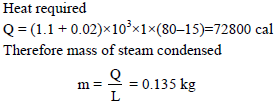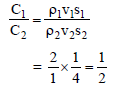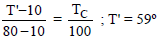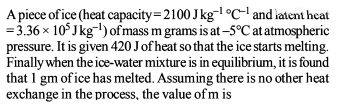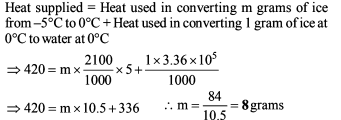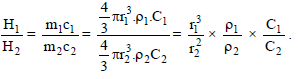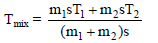Chapter Test: Thermal Properties of Matter - 1 JEE Physics Free MCQs
MCQ Practice Test & Solutions: Chapter Test: Thermal Properties of Matter - 1 (30 Questions)
You can prepare effectively for JEE Physics for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Chapter Test: Thermal Properties of Matter - 1". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Liquids A and B are at 30ºC and 20ºC. When mixed in equal masses, the temperature of the mixture is found to be 26ºC. Their specific heats are of ratio -
Detailed Solution: Question 1
The net force between two nucleons 1 fm apart is F1if both are protons, F2 if both are neutrons, and F3 if one is a neutron and the other is a proton -
Detailed Solution: Question 2
A liquid is being converted into vapours at its bp; the specific heat of liquid at this temperature will be -
Detailed Solution: Question 3
The ratio of densities of two substance is 2 : 3 and their specific heats are in the ratio 3 : 4. The ratio of their thermal capacities for unit volume is -
Detailed Solution: Question 4
Boiling water is changing into steam. At this stage the specific heat of water is-
Detailed Solution: Question 5
300 g of water at 25°C is added to 100g of ice at 0°C. The amount of ice melts is -
Detailed Solution: Question 6
22 gm of CO2 at 27°C is mixed with 17 gm of O2 at 37°C. If both gases are considered as ideal kinetic theory gases then the temperature of the mixture is-
Detailed Solution: Question 7
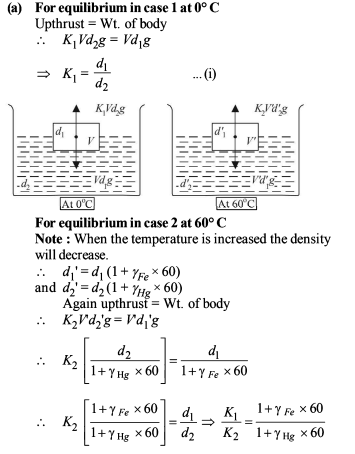
A piece of iron of mass m = 325 g is placed in a calorimeter filled with thawing ice. The amount of ice that will melt by the time thermal equilibrium is reached is (n.4) × 101 g where n is a single digit number. If the volume of the piece of iron being lowered into the calorimeter is V = 48 cm3. The density of iron at 0ºC is d0 = 6.8 g/cm3 , its thermal capacity C = 0.12 cal/g ºC and the coefficient of volume expansion of iron is γ = 3.3 × 10–5/ ºC. Find the value of n.
Detailed Solution: Question 8
Detailed Solution: Question 9
The molar heat capacity of a certain substance varies with temperature according to the given equation, C = 27.2 + (4 × 10–3) T. The heat necessary to change the temperature of 2 mol. of the substance from 300K to 700K is -
Detailed Solution: Question 10
4 gms of steam at 100 ºC is added to 20 gms of water at 46 ºC in a container to negligible mass. Assuming no heat is lost to surrounding, the mass of water in container at thermal equilibrium is. Latent heat of vaporisation = 540 cal/gm. Specific heat of water = 1 cal/gm ºC -
Detailed Solution: Question 11
Detailed Solution: Question 12
A calorimeter of negligible heat capacity contains 100 gm water at 40°C. The water cools to 35°C in 5 min.. If water is now replaced by a liquid of same volume as that of water at same initial temperature it cools to 35°C in 2 min. Given sp. heats of water and liquid are 4200 J/kg-°C and 2100 J/kg-°C respectively. Find the density of liquid give answer in….× 100 kg/m3. [Assume Newton law of cooling is applicable]
Detailed Solution: Question 13
Detailed Solution: Question 14
2 kg of ice at – 20 ºC is mixed with 5 kg of water at 20ºC in an insulating vessel having a negligible heat capacity. Calculate the final mass of water remaining in the container. It is given that the specific heats of water and ice are 1 kcal/kg/ºC and 0.5 kcal/kg/ºC while the latent heat of fusion of ice is 80 kcal/kg -
Detailed Solution: Question 15
2 kg ice at – 20ºC is mixed with 5 kg water at 20ºC in an insulating vessel having negligible heat capacity. Calculate the final mass of water remaining in container.
Given sp. heat water = 4.186 kJ K–1 kg–1
sp. heat Ice = 2.092 kJ K–1 kg–1
Latent heat of fusion of ice = 334.7 kJ Kg–1
Detailed Solution: Question 16
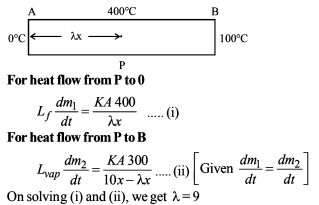
Ice at 0ºC is added to 200gm of water initially at 70°C in a vacuum flask. When 50 gm of ice has been added and has all melted, the temperature of flask and contents is 40ºC, When a further 80 gm of ice is added and has all melted, the temperature of whole becomes 10ºC. Neglecting heat lost to surroundings the latent heat of fusion of ice is :
Detailed Solution: Question 17
Steam at 100ºC is passed into 1.1 kg of water contained in a calorimeter of water equivalent 0.02 kg at 15ºC till the temperature of the calorimeter and its contents rises to 80ºC. The mass of the steam condensed in kg is -
Detailed Solution: Question 18
If two sphare having same radius but the ratio of their specific heat is 2 : 1 and the ratio of their density is 1 : 4 then the ratio of their heat capacity is -
Detailed Solution: Question 19
The steam point and ice point of a mercury thermometer are marked as 80º and 10º. At what temperature on centigrade scale the reading of this thermometer will be 59º ?
Detailed Solution: Question 20
Detailed Solution: Question 21
Two spheres A and B have diameters in the ratio 1 : 2, densities in the ratio 2 : 1 and specific heats in the ratio 1 : 3; find the ratio of their thermal capacities -
Detailed Solution: Question 22
50 gm of ice at 0ºC is mixed with 50 gm of water at 80ºC. The final temperature of mixture is -
Detailed Solution: Question 23
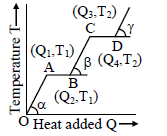
The accompanying graph shows the variation of temperature (T) of one kilogram material with heat (Q) supplied to it. At O, the substance is in solid state. Which of the following interpretations from the graph is correct ?

Detailed Solution: Question 24
In two colorimeters we poured 200 gm of water each at temperature of + 30ºC and +40ºC. From the 'hot' calorimeter 50 gm of water, is poured into 'cold' calorimeter and stirred. Then, from 'cold' colorimeter 50 gm of water is poured into 'hot' and again stirred . How many times from starting do you have to poured the same portion of water back and forth so that temperature difference between water in colorimeters becomes less than 3ºC? Heat loss during transfer and heat capacity of calorimeters is neglected.
Detailed Solution: Question 25
Four cylindrical rods of different radii and lengths are used to connect two heat reservoirs at fixed temperatures t1 and t2 respectively. From the following pick out the rod which will conduct the maximum quantity of heat:
Detailed Solution: Question 26
The temperature for which the reading on Celsius and Fahrenheit scales are identical is
Detailed Solution: Question 27
Which phase of matter has maximum value of temperature coefficient of cubical expansion?
Detailed Solution: Question 28
The coefficient of liner expansion of a cubical crystal along three mutually perpendicular direction is  and
and  . What is the coefficient of cubical expansion of crystal?
. What is the coefficient of cubical expansion of crystal?
 and
and  . What is the coefficient of cubical expansion of crystal?
. What is the coefficient of cubical expansion of crystal?Detailed Solution: Question 29
Water contract on heating between the temperatures
Detailed Solution: Question 30
258 videos|856 docs|206 tests |