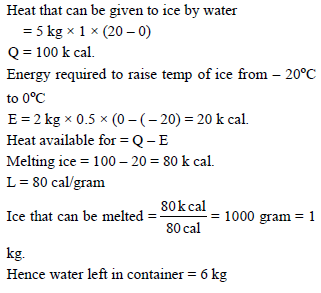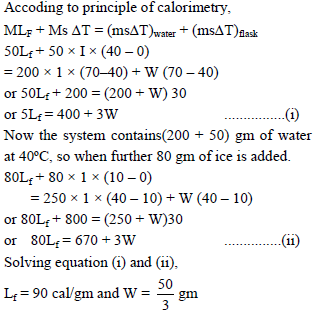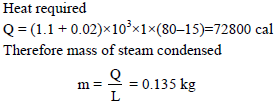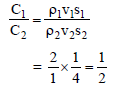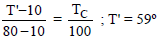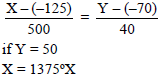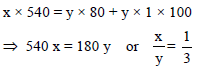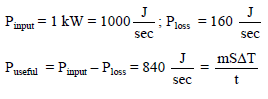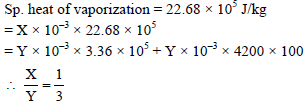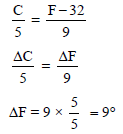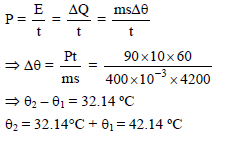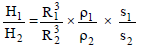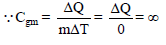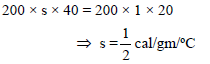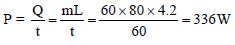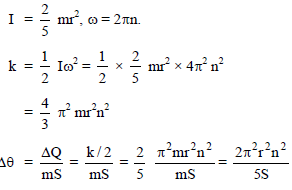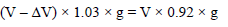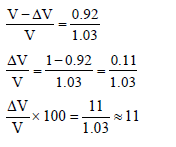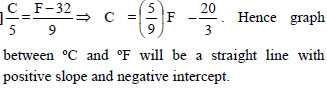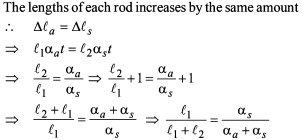Chapter Test: Thermal Properties of Matter - 2 JEE Physics Free MCQs
MCQ Practice Test & Solutions: Chapter Test: Thermal Properties of Matter - 2 (30 Questions)
You can prepare effectively for JEE Physics for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Chapter Test: Thermal Properties of Matter - 2". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
2 kg ice at – 20ºC is mixed with 5 kg water at 20ºC in an insulating vessel having negligible heat capacity. Calculate the final mass of water remaining in container.
Given sp. heat water = 4.186 kJ K–1 kg–1
sp. heat Ice = 2.092 kJ K–1 kg–1
Latent heat of fusion of ice = 334.7 kJ Kg–1
Detailed Solution: Question 1
Ice at 0ºC is added to 200gm of water initially at 70°C in a vacuum flask. When 50 gm of ice has been added and has all melted, the temperature of flask and contents is 40ºC, When a further 80 gm of ice is added and has all melted, the temperature of whole becomes 10ºC. Neglecting heat lost to surroundings the latent heat of fusion of ice is :
Detailed Solution: Question 2
Steam at 100ºC is passed into 1.1 kg of water contained in a calorimeter of water equivalent 0.02 kg at 15ºC till the temperature of the calorimeter and its contents rises to 80ºC. The mass of the steam condensed in kg is -
Detailed Solution: Question 3
If two sphare having same radius but the ratio of their specific heat is 2 : 1 and the ratio of their density is 1 : 4 then the ratio of their heat capacity is -
Detailed Solution: Question 4
The steam point and ice point of a mercury thermometer are marked as 80º and 10º. At what temperature on centigrade scale the reading of this thermometer will be 59º ?
Detailed Solution: Question 5
On an X temperature scale, water freezes at –125ºX and boils at 375ºX. On a Y temperature scale water freezes at –70ºY and boils at –30ºY The value of temperature on X-scale equal to the temperature of 50ºY on Y-scale is ..............ºX.
Detailed Solution: Question 6
If x grams of steam at 100ºC becomes water at 100ºC which converts y grams of ice at 0ºC into water at 100ºC, then the ratio x/y will be –
Detailed Solution: Question 7
The weight of a hammer is 0.5 kg. It falls at the ratio of 10 strokes per minute on a piece of lead of mass 0.03 kg at 25ºC with a velocity 1.0 m/s. The number of strokes of hammer required to melt the lead piece will be (melting point of lead = 325ºC, specific heat of lead = 0.03 cal/gm-ºC and latent heat = 6 kcal/kg) -
Detailed Solution: Question 8
A beaker is completely filled with water at 4ºC. It will overflow -
Detailed Solution: Question 9
A kettle with 2 litre water at 27°C is heated by operating coil heater of power 1 kW. The heat is lost to the atmosphere at constant rate 160J/sec, when its lid is open. In how much time will water heated to 77°C with the lid open ? (specific heat of water = 4.2 kJ/ kg)
Detailed Solution: Question 10
In an energy recycling process, X g of steam at 100°C becomes water at 100°C which converts Y g of ice at 0°C into water at 100°C. The ratio of X/Y will be -
Detailed Solution: Question 11
A temperature difference of 5°C on Celsius scale corresponding to the following temperature difference in the Fahrenheit scale -
Detailed Solution: Question 12
An electrically heated coil is immersed in a calorimeter containing 360 gm of water at 10ºC. The coil consumes energy at the rate of 90W. The water equivalent of calorimeter & coil is 40 gm. The temperature of water after 10 minutes is-
Detailed Solution: Question 13
Assertion (A) : Melting of solid causes no change in internal energy
Reason (R) : Specific latent heat is the heat required to melt a unit mass of solid
Detailed Solution: Question 14
Two liquids A and B are at 32°C and 24°C. When mixed in equal masses the temperature of the mixture is found to be 28°C. Their specific heats are in the ratio of -
Detailed Solution: Question 15
A liquid is being converted into vapours at its bp; the specific heat of liquid at this temperature will be –
Detailed Solution: Question 16
200 g of a solid ball at 20ºC is dropped in an equal amount of water at 80ºC. The resulting temperature is 60ºC. This means that specific heat of solid is -
Detailed Solution: Question 17
Heat required to melt 1 gm of ice is 80cal. A man melts 60 gms of ice by chewing in 1 min. His power is -
Detailed Solution: Question 18
A metal sphere of radius r and specific heat S is rotated about an axis passing through its centre at a speed of n rotations per second. It is suddenly stopped and 50 % of its energy is used in increasing its temperature. Then the rise in temperature of the sphere is -
Detailed Solution: Question 19
An iceberg is floating partially immersed in sea water. The density of sea water is 1.03 g cm–3 and that of ice is 0.92 g cm–3. The approximate percentage of total volume of iceberg above the level of sea water is -
Detailed Solution: Question 20
Which of the curves in figure represents the relation between Celsius and Fahrenheit temperature-
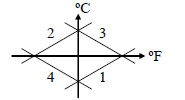

Detailed Solution: Question 21
A block of mass 2 kg is kept at temperature 200ºC is put into a calorimeter of water equivalent 40 gm contain 960 gm of water at 40ºC. The resulting temperature of mixture is (Sp. heat of block = 0.1 Cal/gm-ºC)
Detailed Solution: Question 22
Detailed Solution: Question 23
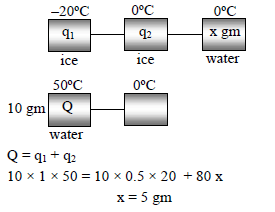
When 10 gm of ice at –20 ºC is mixed with 10 gm of water at 50 ºC, the amount of ice melted is -
Detailed Solution: Question 24
A calorimeter (water equivalent 50 gm) contains 20 gm ice at – 20ºC. Find the amount of steam at 100ºC require to just melt all the ice.(Sw= 1 cal/gm-ºC, Sice = 0.5 cal/gm-ºC, Lf = 80 cal/gm, Lv = 540 cal/gm)
Detailed Solution: Question 25
Four cylindrical rods of different radii and lengths are used to connect two heat reservoirs at fixed temperatures t1 and t2 respectively. From the following pick out the rod which will conduct the maximum quantity of heat:
Detailed Solution: Question 26
A steel tape is calibrated at 20° C. A piece of wood is being measured by steel tape at 10°C and reading is 30 cm on the tape. The real length of the wood is:
Detailed Solution: Question 27
The coefficient of liner expansion of a cubical crystal along three mutually perpendicular direction is  and
and  . What is the coefficient of cubical expansion of crystal?
. What is the coefficient of cubical expansion of crystal?
 and
and  . What is the coefficient of cubical expansion of crystal?
. What is the coefficient of cubical expansion of crystal?Detailed Solution: Question 28
Relationship for converting the Fahrenheit and Celsius scales is
Detailed Solution: Question 29
When water is heated from 0° C to 20° C its volume:
Detailed Solution: Question 30
258 videos|856 docs|206 tests |