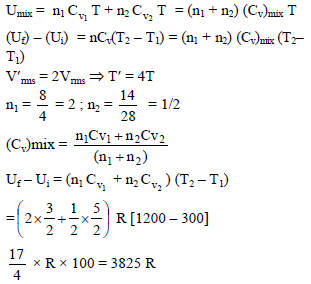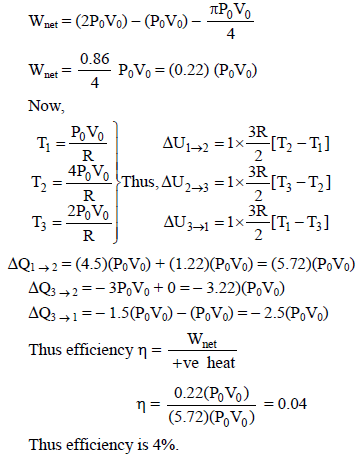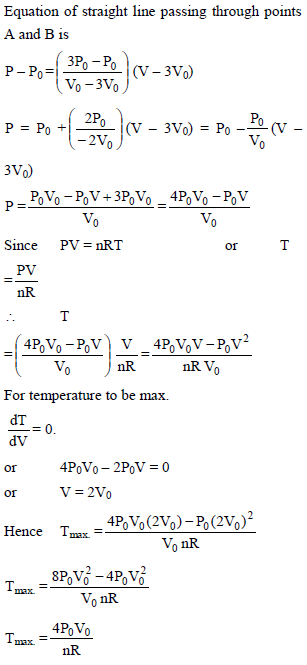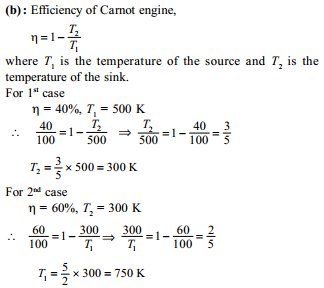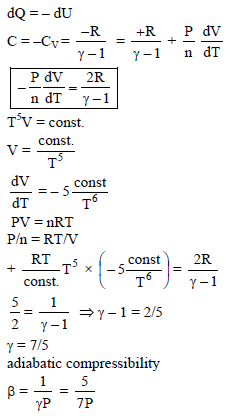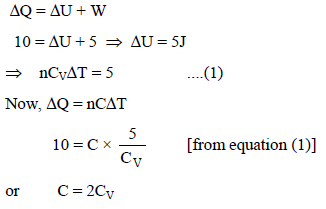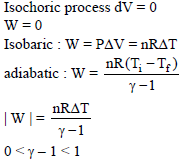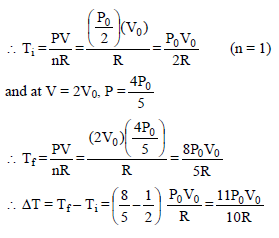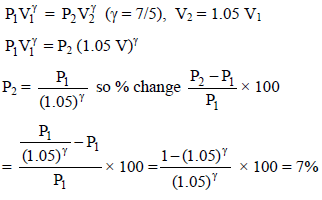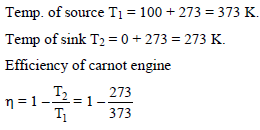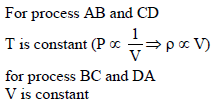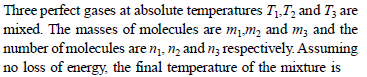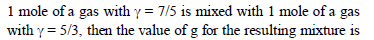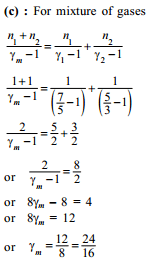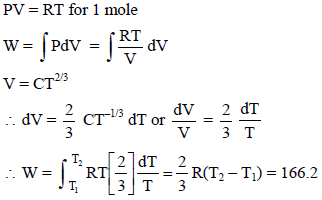Chapter Test: Thermodynamics -2 JEE Physics Free MCQs
MCQ Practice Test & Solutions: Chapter Test: Thermodynamics -2 (30 Questions)
You can prepare effectively for JEE Physics for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Chapter Test: Thermodynamics -2". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
Detailed Solution: Question 2
A mixture of 8gm of helium and 14gm of nitrogen is enclosed in a vessel of constant volume at 300K. The quantity of heat absorbed by the mixture to double the root mean velocity of its molecules is –
(R = universal gas constant )
Detailed Solution: Question 3
One mole of ideal monoatomic gas is taken along a cyclic process as shown in the figure. Process
1 → 2 shown is 1/4th part of a circle as shown by dotted line process 2 → 3 is isochoric while
3 → 1 is isobaric. If efficiency of the cycle is n% where n is an integer. Find n.
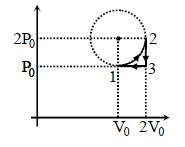
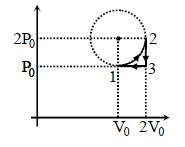
Detailed Solution: Question 4
Detailed Solution: Question 5
n moles of an ideal gas undergoes a process A to B as shown. Maximum temperature of gas during the process is –
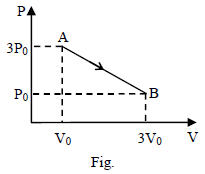

Detailed Solution: Question 6
Detailed Solution: Question 7
An ideal gas is expanded so that amount of heat given is equal to the decrease in internal energy. The gas undergoes the process TV1/5 = constant. The adiabatic compressibility of gas when pressure is P, is –
Detailed Solution: Question 8
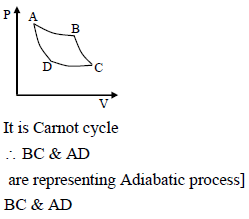
The pressure volume graph of an ideal gas cycle is shown in the fig. The adiabatic process is described by
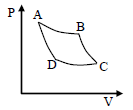

Detailed Solution: Question 9
In a given process of an ideal gas, dW = 0 and dQ < 0. Then for the gas –
Detailed Solution: Question 10
The molar heat capacity for the process is xR, where R is gas constant, when 10 J of heat added to a monoatomic ideal gas, then gas performs a work of 5 J on its surrounding, then x is.
Detailed Solution: Question 11
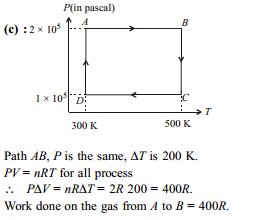
The figure shows two isotherms at temperatures T1 and T2. A gas is taken from one isotherm to another isotherm through different processes. Then change in internal energy ΔU has relation -
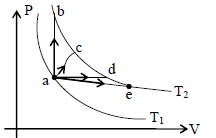

Detailed Solution: Question 12
Detailed Solution: Question 13
For one complete cycle of a thermodynamic process on a gas as shown in the P-V diagram. Which of the following is correct ?
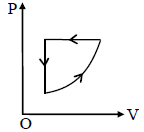

Detailed Solution: Question 14
Detailed Solution: Question 15
1 mole of a monoatomic and 2 mole of diatomic gas are mixed, Now the resulting gas is taken through a process in which molar heat capacity was found 3R. Polytropic constant in the process is -
Detailed Solution: Question 16
For an ideal gas graph is shown for three processes. Processes 1, 2, and 3 are respectively –
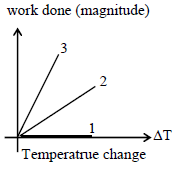

Detailed Solution: Question 17
Detailed Solution: Question 18
In an adiabatic expansion of air the volume increases by 5%. What is the percentage change in pressure ? [(1.05)7/5 = 1.07]
Detailed Solution: Question 19
A carnot engine works between ice point and steam point. It is desired to increase efficiency by 20%, by changing temperature of sink to –
Detailed Solution: Question 20
Pressure versus temperature graph of an ideal gas is as shown in figure corresponding density (ρ) versus volume (V) graph will be -
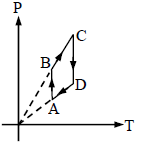

Detailed Solution: Question 21
Detailed Solution: Question 22
Detailed Solution: Question 23
Find the amount of work done to increase the temperature of one mole of an ideal gas by 30ºC if it is expanding under the condition V ∝ T2/3 -
Detailed Solution: Question 24
Detailed Solution: Question 25
For proper utilization of exergy, it is desirable to make first law efficiency ____ and the source and use temperatures should ____.
Detailed Solution: Question 26
If the door of refrigerator is left open inside a closed room, what would happen to the temperature of the room?
Detailed Solution: Question 27
Refrigerator transfers heat from the cold cooling coils to warm surroundings, which law of thermodynamics favour this process
Detailed Solution: Question 28
What amount of work is needed to freeze 1 gm of water at 0˚C in a refrigerator .The temperature of surroundings is 320 K?
Detailed Solution: Question 29
The coefficient of performance of a refrigerator is given by
Detailed Solution: Question 30
258 videos|856 docs|206 tests |