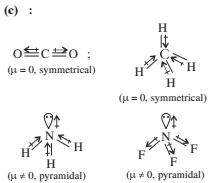
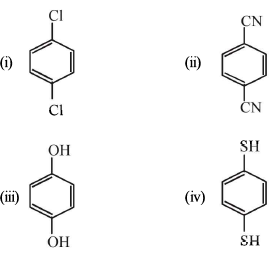
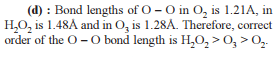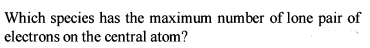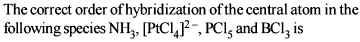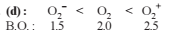Chemical Bonding - 1 - Free MCQ Test with solutions for JEE Chemistry
MCQ Practice Test & Solutions: Chemical Bonding - 1 (30 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Chemical Bonding - 1". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
A : tetracyanomethane B : Carbondioxide
C : Benzene D : 1, 3-buta-di-ene
Ratio of σ and π bond is in order:
Detailed Solution: Question 1
In 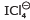 , the shape is square planer. The number of bond pair-lone pair repulsion at 90° are :
, the shape is square planer. The number of bond pair-lone pair repulsion at 90° are :
Detailed Solution: Question 2
Detailed Solution: Question 3
Detailed Solution: Question 4
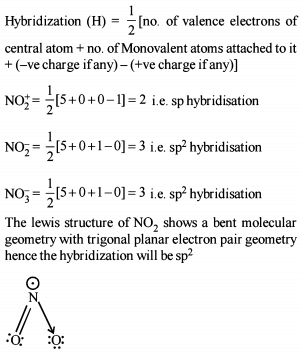
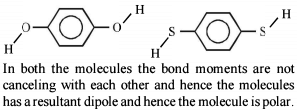
Detailed Solution: Question 5
Detailed Solution: Question 6
Detailed Solution: Question 7
Detailed Solution: Question 8
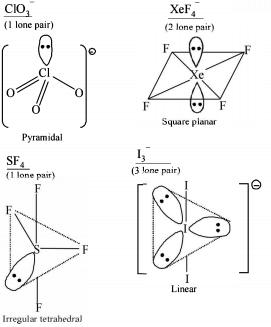
Detailed Solution: Question 9
Detailed Solution: Question 10
Detailed Solution: Question 11
Detailed Solution: Question 12
Detailed Solution: Question 13
Detailed Solution: Question 14
Detailed Solution: Question 15
Among the following, the molecule expected to be stabilized by anion formation is :
C2, O2, NO, F2
C2, O2, NO, F2
Detailed Solution: Question 16
Detailed Solution: Question 17
Detailed Solution: Question 18
Detailed Solution: Question 19
Detailed Solution: Question 20
Detailed Solution: Question 21
Detailed Solution: Question 22
Detailed Solution: Question 23
Detailed Solution: Question 24
Detailed Solution: Question 25
Detailed Solution: Question 26
Detailed Solution: Question 27
Detailed Solution: Question 28
Detailed Solution: Question 29
Detailed Solution: Question 30
335 videos|697 docs|300 tests |