CUET UG Chemistry: Mock Test - 5 Free Online Test 2026
Full Mock Test & Solutions: Chemistry: CUET Mock Test - 5 (50 Questions)
You can boost your CUET 2026 exam preparation with this Chemistry: CUET Mock Test - 5 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of CUET 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Total Questions: 50
- - Analysis: Detailed Solutions & Performance Insights
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
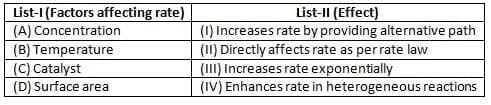
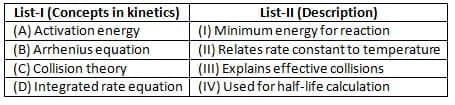
Match list I with list II
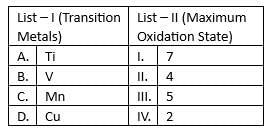
Choose the correct answer from the options given below:

Choose the correct answer from the options given below:
Detailed Solution: Question 1
The following statements are related to the rate law and rate constant of a chemical reaction:
(A) The rate constant k is independent of the concentration of reactants
(B) The unit of rate constant for a first-order reaction is s-1
(C) The rate law for a reaction can be determined theoretically from the balanced chemical equation
(D) The order of a reaction with respect to a reactant can be a fraction
Choose the correct answer from the options given below:
Detailed Solution: Question 2
The following statements describe the half-life of a reaction:
(A) The half-life of a first-order reaction is independent of the initial concentration of the reactant
(B) The half-life of a zero-order reaction is directly proportional to the initial concentration
(C) The half-life of a second-order reaction is independent of the initial concentration
(D) For a first-order reaction, t(1/2) = 0.693/k
Choose the correct answer from the options given below:
Detailed Solution: Question 3
Detailed Solution: Question 4
Detailed Solution: Question 5
Arrange the following transition elements in decreasing order of their maximum oxidation state:
(A) V (Vanadium)
(B) Sc (Scandium)
(C) Cr (Chromium)
(D) Mn (Manganese)
Choose the correct order:
Detailed Solution: Question 6
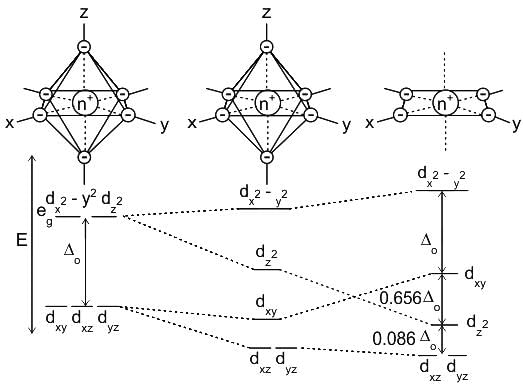
Arrange the following ligands in increasing order of their crystal field splitting energy (Δ) as per the spectrochemical series:
(A) NH₃
(B) Cl⁾
(C) CN⁾
(D) H₂O
Choose the correct order:
Detailed Solution: Question 7
The metal from first transition series having positive 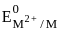 value :
value :
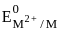 value :
value :Detailed Solution: Question 8
Detailed Solution: Question 9
According to CFT (crystal field theory) [Ni(CN)4]2- has ___________.
Detailed Solution: Question 10
Consider the following statements regarding electrochemical cells:
(A) A higher standard electrode potential indicates a greater tendency to gain electrons.
(B) A negative standard electrode potential indicates a higher tendency to gain electrons.
(C) In a galvanic cell, the electrons flow from the anode to the cathode through the external circuit.
(D) In an electrolytic cell, the cathode is the site of oxidation.
Choose the correct statements:
(A) A higher standard electrode potential indicates a greater tendency to gain electrons.
(B) A negative standard electrode potential indicates a higher tendency to gain electrons.
(C) In a galvanic cell, the electrons flow from the anode to the cathode through the external circuit.
(D) In an electrolytic cell, the cathode is the site of oxidation.
Choose the correct statements:
Detailed Solution: Question 11
Consider the following statements regarding electrode potentials:
(A) Standard electrode potential is always measured under standard conditions (1 M concentration, 1 atm pressure, 25°C).
(B) The standard electrode potential of a cell is the sum of the cathode and anode potentials.
(C) A positive electrode potential indicates that the electrode is more likely to lose electrons.
(D) The standard electrode potential is a measure of the tendency of a species to gain electrons.
Choose the correct statements:
(A) Standard electrode potential is always measured under standard conditions (1 M concentration, 1 atm pressure, 25°C).
(B) The standard electrode potential of a cell is the sum of the cathode and anode potentials.
(C) A positive electrode potential indicates that the electrode is more likely to lose electrons.
(D) The standard electrode potential is a measure of the tendency of a species to gain electrons.
Choose the correct statements:
Detailed Solution: Question 12
Consider the following statements about redox reactions:
(A) In a redox reaction, oxidation involves the loss of electrons.
(B) In a redox reaction, reduction involves the gain of electrons.
(C) A reducing agent is a substance that gains electrons and is reduced.
(D) An oxidizing agent is a substance that gains electrons and is reduced.
Choose the correct statements:
(A) In a redox reaction, oxidation involves the loss of electrons.
(B) In a redox reaction, reduction involves the gain of electrons.
(C) A reducing agent is a substance that gains electrons and is reduced.
(D) An oxidizing agent is a substance that gains electrons and is reduced.
Choose the correct statements:
Detailed Solution: Question 13
Consider the following statements about reaction rate:
(A) The rate of a reaction is always constant for a given temperature.
(B) The rate of a reaction is defined as the change in concentration of reactants or products per unit time.
(C) The rate of reaction increases with temperature.
(D) The rate of reaction is unaffected by the concentration of reactants.
Choose the correct statements:
(A) The rate of a reaction is always constant for a given temperature.
(B) The rate of a reaction is defined as the change in concentration of reactants or products per unit time.
(C) The rate of reaction increases with temperature.
(D) The rate of reaction is unaffected by the concentration of reactants.
Choose the correct statements:
Detailed Solution: Question 14
A solution of Na2SO4 in water is electrolysed using Pt electrodes.The products at the cathode and anode are respectively -
Detailed Solution: Question 15
For the reaction system: 2NO(g) + O2(g) → 2NO2(g) volume is suddenly reduced to half its value by increasing the pressure on it. If the reaction is of first order with respect to O2 and second order with repect to NO, the rate of reaction will –
[AIEEE-2003]
Detailed Solution: Question 16
Rate of reaction can be expressed by Arrhenius equation as k = Ae–E/RT , In this equation, E represents
[AIEEE 2006]
Detailed Solution: Question 17
Detailed Solution: Question 18
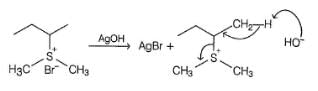
Which of the following gives 1-butene as the major product most easily on heating with AgOH?
Detailed Solution: Question 19
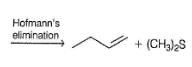
1-butene would be formed most easily in the following reaction when X is
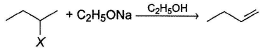
Detailed Solution: Question 20
Which of the following compound is most likely to follow E1 cb mechanism when treated with C2H5ONa in ethanol?
Detailed Solution: Question 21
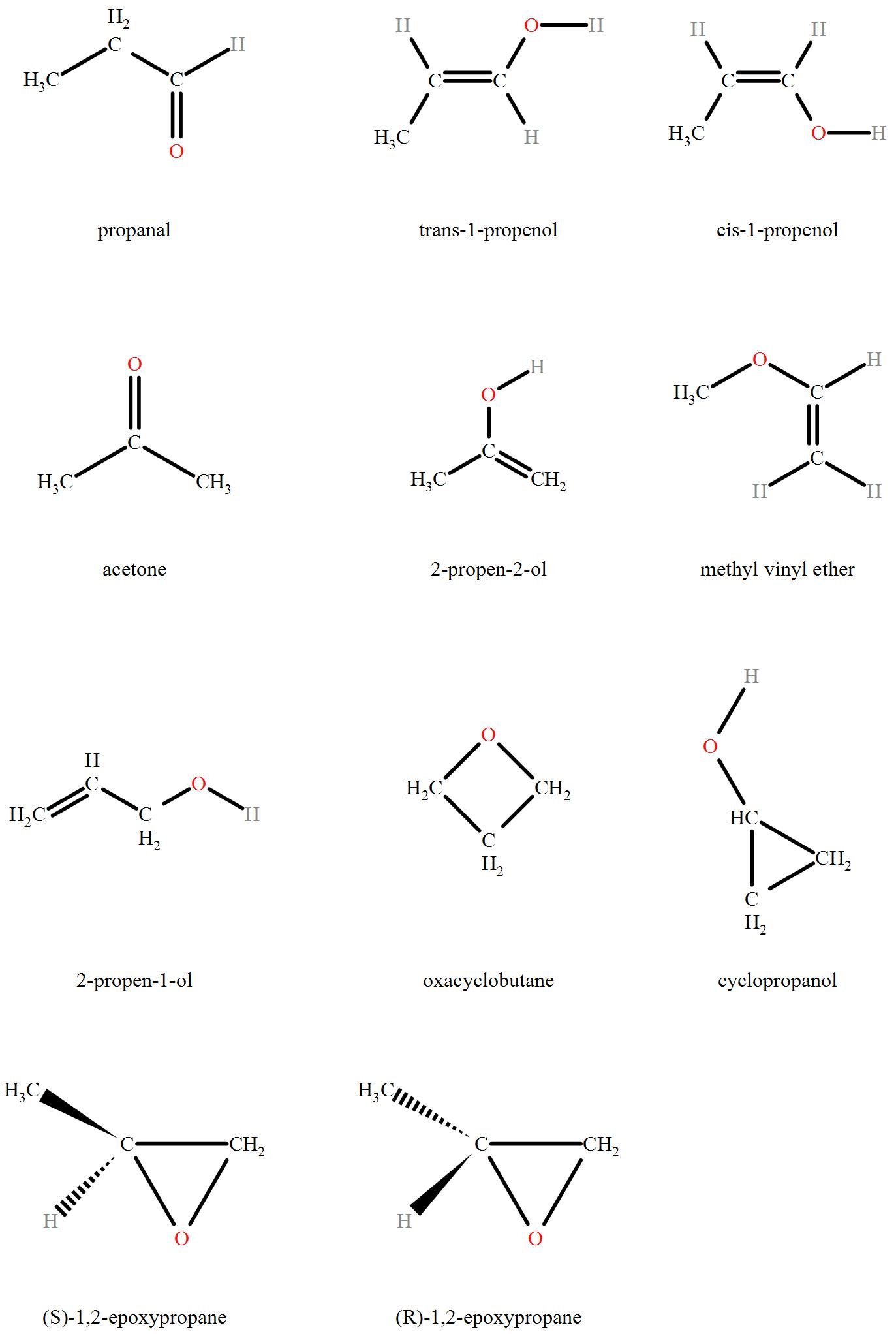
How many structural isomers can compound with molecular formula ‘C3H6O’ have?
Detailed Solution: Question 22
Detailed Solution: Question 23
Detailed Solution: Question 24
Carbonyl compounds when oxidized easily to form carboxylic group involves
Detailed Solution: Question 25
Detailed Solution: Question 26
Which of the following polymer is stored in the liver of animals?
Detailed Solution: Question 27
Detailed Solution: Question 28
Detailed Solution: Question 29
Detailed Solution: Question 30
41 docs|160 tests |



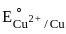 value in 3d series.
value in 3d series.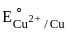 = 0.34 V
= 0.34 V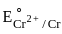 = –0.90 V
= –0.90 V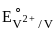 = –1.18 V
= –1.18 V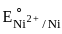 = = –0.25 V
= = –0.25 V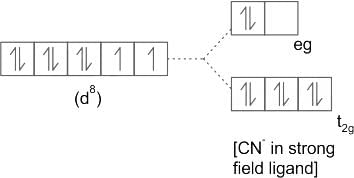

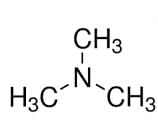 is a tertiary amine with IUPAC name:
is a tertiary amine with IUPAC name: