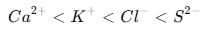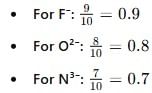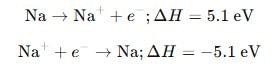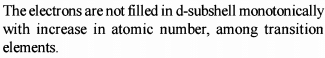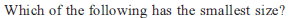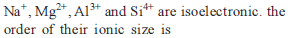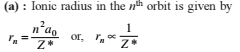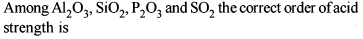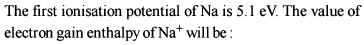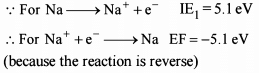Classification of Elements and Periodicity in Properties - 1 - Free MCQ
MCQ Practice Test & Solutions: Classification of Elements and Periodicity in Properties - 1 (30 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Classification of Elements and Periodicity in Properties - 1". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which is the correct order of ionic sizes (At. No. : Ce = 58, Sn = 50, Yb = 70 and Lu = 71) [AIEEE-2002]
Detailed Solution: Question 1
The reduction in atomic size with increase in atomic number is a characteristic of elements of - [AIEEE-2003]
Detailed Solution: Question 2
The atomic numbers of vanadium (V). Chromium (Cr), manganese (Mn) and iron (Fe) respectively 23, 24, 25 and 26. Which one of these may be expected to have the higher second ionization enthalpy ? [AIEEE-2003]
Detailed Solution: Question 3
Which one of the following sets of ions represents the collection of isoelectronic species ? [AIEEE-2004]
Detailed Solution: Question 4
lanthanoid contraction is caused due to - [AIEEE-2006]
Detailed Solution: Question 5
The increasing order of the ionic radii of the given isoelectronic species is:
Detailed Solution: Question 6
The increasing order of the ionic radii of the given isoelectronic species is:
Detailed Solution: Question 7
Identify the correct order of the size of the following:
Detailed Solution: Question 8
Pauling’s electronegativity values for elements are useful in predicting
Detailed Solution: Question 9
Amongst the elements with the following electronic configurations, which one of them may have the highest ionisation energy?
Detailed Solution: Question 10
The correct order of radii is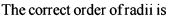
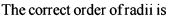
Detailed Solution: Question 11
Choose the correct order of the following:
Detailed Solution: Question 12
Which has the most stable +2 oxidation state?
Detailed Solution: Question 13
What is the value of electron gain enthalpy of Na⁺ if IE₁ of Na = 5.1 eV?
Detailed Solution: Question 14
Atomic radii of fluorine and neon in Ångstrom units are respectively given by:
Detailed Solution: Question 15
Detailed Solution: Question 16
Detailed Solution: Question 17
Detailed Solution: Question 18
Detailed Solution: Question 19
Detailed Solution: Question 20
Detailed Solution: Question 21
Detailed Solution: Question 22
Detailed Solution: Question 23
Detailed Solution: Question 24
Detailed Solution: Question 25
Detailed Solution: Question 26
Detailed Solution: Question 27
Detailed Solution: Question 28
Detailed Solution: Question 29
Detailed Solution: Question 30
332 videos|699 docs|300 tests |