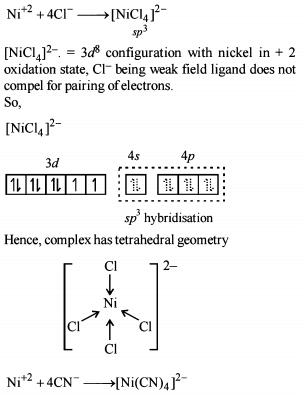
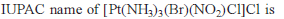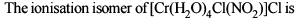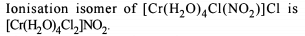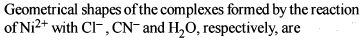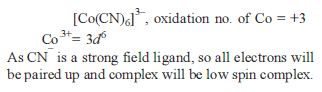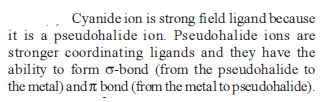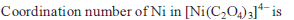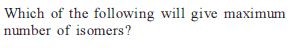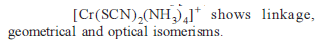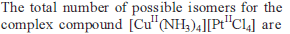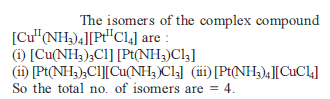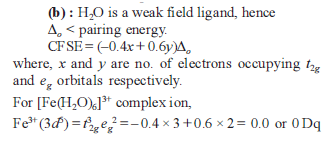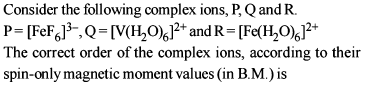Co-ordination Compounds - 1 - Free MCQ Test with solutions for JEE Chemistry
MCQ Practice Test & Solutions: Co-ordination Compounds - 1 (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Co-ordination Compounds - 1". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 40 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
In the complexes [Fe(H2O)6]3+, [Fe(CN)6]3+, [Fe(C2O4)3]3_ and [FeCl6]3_, more stability is shown by -
Detailed Solution: Question 1
The value of the 'spin only' magnetic moment for one of the following configurations is 2.84 BM . The correct one is –
[AIEEE-2005]
Detailed Solution: Question 2
In which of the following octahedral complexes of Co (at. no. 27), will the magnitude of D0 be the highest ?
Detailed Solution: Question 3
Detailed Solution: Question 4
Detailed Solution: Question 5
Detailed Solution: Question 6
Detailed Solution: Question 7
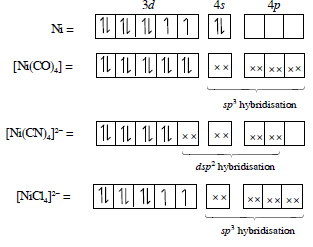
Detailed Solution: Question 8
Detailed Solution: Question 9
Detailed Solution: Question 10
Detailed Solution: Question 11
Detailed Solution: Question 12
Detailed Solution: Question 13
Detailed Solution: Question 14
Detailed Solution: Question 15
Detailed Solution: Question 16
Detailed Solution: Question 17
Detailed Solution: Question 18
Detailed Solution: Question 19
Detailed Solution: Question 20
335 videos|699 docs|300 tests |