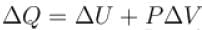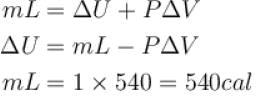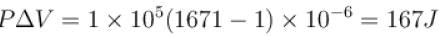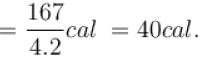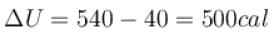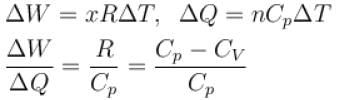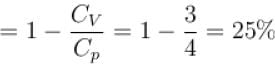First Law Of Thermodynamics MCQ Level – 2 (Part - 1) Physics MCQs & solutions
MCQ Practice Test & Solutions: First Law Of Thermodynamics MCQ Level – 2 (Part - 1) (10 Questions)
You can prepare effectively for Physics Topic wise Tests for IIT JAM Physics with this dedicated MCQ Practice Test (available with solutions) on the important topic of "First Law Of Thermodynamics MCQ Level – 2 (Part - 1)". These 10 questions have been designed by the experts with the latest curriculum of Physics 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 45 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
One mole of an ideal gas expands adiabatically from temperature T1 to temperature T2. The work done by the gas is
Select one:
Select one:
Detailed Solution: Question 1
The pressure-volume graph of an ideal gas cycle consisting of isothermal and adiabatic process is shown in the figure. The adiabatic process is described by
Select one:

Select one:
Detailed Solution: Question 2
A water fall is 168m high. Assuming that half the kinetic energy of the falling water gets converted into heat, the rise in temperature of water is approximately (take g = 10m/s2)
Detailed Solution: Question 3
One gram of water on evaporation at atmospheric pressure forms 1671cm3 of steam. Heat of vaporization at this pressure is 540 cal/g. Calculate the increase in the internal energy. (Atmospheric pressure = 1×105N/m2)
Select one:
Detailed Solution: Question 4
A sample of gas expands from volume V1 to V2. The amount of work done by the gas in greatest when the expansion is.
Select one:
Select one:
Detailed Solution: Question 5
One gram of water on evaporation at atmospheric pressure forms 1671cm3 of steam. Heat of vaporization at this pressure is 540 cal/g. The increase in internal energy is
Select one:
Select one:
Detailed Solution: Question 6
Heat capacity of a substance is infinite. It means :
Select one:
Select one:
Detailed Solution: Question 7
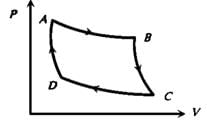
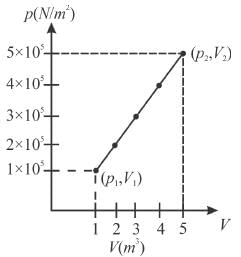
A system changes from the state (p1, V1 ) to the state (p2, V2 ) as shown in the figure. The work done by the system is.
Select one:

Select one:
Detailed Solution: Question 8
A body of mass 25kg is dragged on a rough horizontal road for one hour with a speed of 20km/hr.
If the coefficient of friction is 0.5 and half of the heat produced is absorbed by the body, the rise in its temperature is (specific heat of body = 0.1cal/g ºC, g = 10m/s2)
Select one:
If the coefficient of friction is 0.5 and half of the heat produced is absorbed by the body, the rise in its temperature is (specific heat of body = 0.1cal/g ºC, g = 10m/s2)
Select one:
Detailed Solution: Question 9
A gas, for which γ is 4/3, is heated at constant pressure. The percentage of heat supplied used for external work.
Select one:
Select one:
Detailed Solution: Question 10