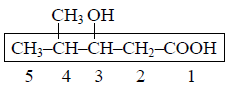General Organic Chemistry (GOC) - 1 - Free MCQ Test with solutions
MCQ Practice Test & Solutions: General Organic Chemistry (GOC) - 1 (30 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "General Organic Chemistry (GOC) - 1". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
Detailed Solution: Question 2
The IUPAC name of the compound
CH₃CH=CHC≡CH is:
CH₃CH=CHC≡CH is:
Detailed Solution: Question 3
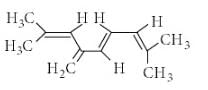
The total number of π-bond electrons in the following structure is:

Detailed Solution: Question 4
Detailed Solution: Question 5
The correct decreasing order of priority for the functional groups of organic compounds in the IUPAC system of nomenclature is
Detailed Solution: Question 6
Detailed Solution: Question 7
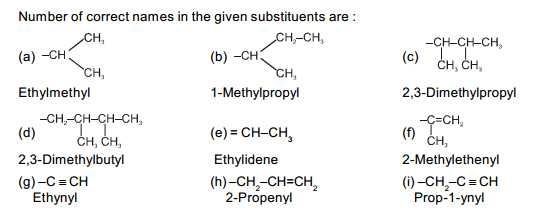
In which of the following compounds is there more than one kind of hybridization (sp, sp², sp³) for carbon?
Detailed Solution: Question 8
The IUPAC name of the compound
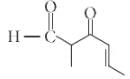 is ______.
is ______.
 is ______.
is ______.Detailed Solution: Question 9
Which of the following is saturated homocyclic compound.
Detailed Solution: Question 10
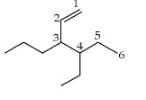
In the structure of 4-Isopropyl-2,4,5-trimethylheptane, number of 10, 20 & 30 H’s are respectively.
Detailed Solution: Question 11
IUPAC nomenclature of the given organic compound will be : (CH3)2C(CH2CH3)CH2CH(Cl)CH3 :
Detailed Solution: Question 12
In allene (C3H4), the type(s) of hybridisation of the carbon atoms is (are) :
Detailed Solution: Question 13
Detailed Solution: Question 14
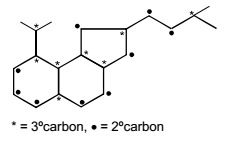
A hydrocarbon (R) has six membered ring in which there is no unsaturation. Two alkyl groups are attached to the ring adjacent to each other. One group has 3 carbon atoms with branching at 1st carbon atom of chain and another has 4 carbon atoms. The larger alkyl group has main chain of three carbon atoms of which second carbon is substituted. Number of 2° carbons in R are :
Detailed Solution: Question 15
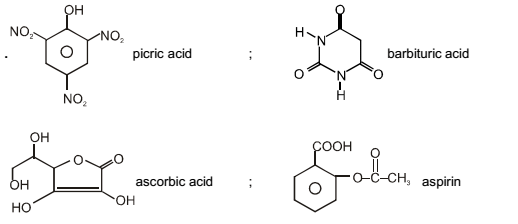
The carboxyl functional group (– COOH) is present in :
Detailed Solution: Question 16
Detailed Solution: Question 17
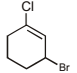
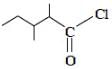
The correct IUPAC name for the compound
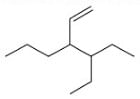 is ______.
is ______.
 is ______.
is ______.Detailed Solution: Question 18
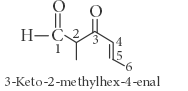
The correct IUPAC name for the compound
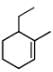 is ______.
is ______.
 is ______.
is ______.Detailed Solution: Question 19
Detailed Solution: Question 20
Detailed Solution: Question 21
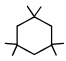
In above compound total number of 2º hydrogen atoms are :

Detailed Solution: Question 22
The IUPAC name of the following compound is -
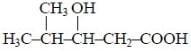
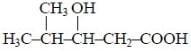
Detailed Solution: Question 23
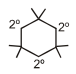
Number of 3º and 2º carbon atoms in the following compound are.
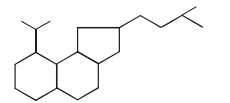

Detailed Solution: Question 24
Which of the following statements is not correct?
Detailed Solution: Question 25
Observe the following compound and write the number of hydrogen atom involved in hyperconjugation ?
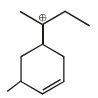

Detailed Solution: Question 26
Which of the following groups cannot participate in resonance with benzene :
Detailed Solution: Question 27
Among the following four compounds:
(i) Phenol
(ii) Methyl phenol
(iii) Meta-nitrophenol
(iv) Para-nitrophenol
The acidity order is:
Detailed Solution: Question 28
Which of the following compounds will not show geometrical isomerism?
Detailed Solution: Question 29
In the compound CH3CH2CH(CH3)CH2CH3, how many primary (1°), secondary (2°), and tertiary (3°) carbon atoms are present?
Detailed Solution: Question 30
335 videos|697 docs|300 tests |



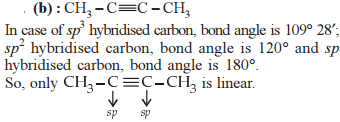







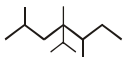
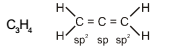

 is ______.
is ______.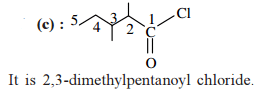
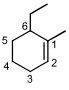
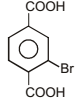
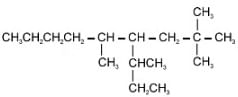 is
is