HOTS Questions: Atoms and Molecules - Free MCQ Test with solutions
MCQ Practice Test & Solutions: HOTS Questions: Atoms and Molecules (25 Questions)
You can prepare effectively for Class 9 Online MCQ Tests for Class 9 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "HOTS Questions: Atoms and Molecules". These 25 questions have been designed by the experts with the latest curriculum of Class 9 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 25 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
Who suggested that if we go on dividing matter, a stage will come when particles obtained can’t be divided further?
Detailed Solution: Question 1
Detailed Solution: Question 2
What information we get from molecular formula
A It represents one molecule of the substance
B It does not tells the name of the substance
C It tells about the type of atoms
D It represents formula mass unit of one substance
A It represents one molecule of the substance
B It does not tells the name of the substance
C It tells about the type of atoms
D It represents formula mass unit of one substance
Detailed Solution: Question 3
Detailed Solution: Question 4
Detailed Solution: Question 5
Detailed Solution: Question 6
Detailed Solution: Question 7
Calculate the number of Mg atoms in 0.024 g of Mg
Detailed Solution: Question 8
Who was the first to use the symbols for elements?
Detailed Solution: Question 9
What according to Dalton’s atomic theory is true among the following-
A. Atom is divisible into Protons, electrons and neutrons
B. Atoms of the same element have different atomic masses.
C. Atoms are the ultimate indivisible particle of matter
D. Atoms of same element have same atomic masses size and chemical properties
A. Atom is divisible into Protons, electrons and neutrons
B. Atoms of the same element have different atomic masses.
C. Atoms are the ultimate indivisible particle of matter
D. Atoms of same element have same atomic masses size and chemical properties
Detailed Solution: Question 10
Which of the following elements are present in Quick lime?
Detailed Solution: Question 11
Statement A: mole is quite often known as chemists dozen
Statement B: the mass of one twelfth (1/12) of the mass of one atom of carbon taken as 1u.
Which of the two statement is true
Statement B: the mass of one twelfth (1/12) of the mass of one atom of carbon taken as 1u.
Which of the two statement is true
Detailed Solution: Question 12
Detailed Solution: Question 13
During a chemical reaction, the sum of the masses of the reactants and products remains unchanged. This is known as
Detailed Solution: Question 14
Detailed Solution: Question 15
How many molecules are present in one gram molecular mass of a substance?
Detailed Solution: Question 16
Detailed Solution: Question 17
Statement A: Atoms can exist independently
Statement B: The law of constant proportions is applicable only to pure chemical compounds.
Which of the two Statements is true?
Statement B: The law of constant proportions is applicable only to pure chemical compounds.
Which of the two Statements is true?
Detailed Solution: Question 18
What is the mass of 0.5 mole of Hydrogen atom
Detailed Solution: Question 19
The atomic mass of calcium is 40 the number of moles in 60 g of calcium are
Detailed Solution: Question 20
What is the name given to the short hand representation of an element?
Detailed Solution: Question 21
Which postulates of Dalton’s atomic theory is the basis of the law of conservation of mass?
Detailed Solution: Question 22
Detailed Solution: Question 23
Detailed Solution: Question 24
Detailed Solution: Question 25
5 docs|319 tests |




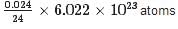 = 6.022 x 1020 atoms.
= 6.022 x 1020 atoms.