HPSC PGT Chemistry Mock Test - 8 Free Online Test 2026
Full Mock Test & Solutions: HPSC PGT Chemistry Mock Test - 8 (100 Questions)
You can boost your HPSC TGT/PGT 2026 exam preparation with this HPSC PGT Chemistry Mock Test - 8 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of HPSC TGT/PGT 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 120 minutes
- - Total Questions: 100
- - Analysis: Detailed Solutions & Performance Insights
- - Sections covered: Part 1: GK/ CA/ Reasoning & Pedagogy, Part 2: Subject
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
Detailed Solution: Question 1
Who among the following was NOT the Governor of Haryana?
Detailed Solution: Question 2
Four word-pairs have been given, out of which three are alike in some manner and one is different. Select the odd one.
Detailed Solution: Question 3
Select the number which can be placed at the sign of the question mark (?) from the given alternatives.
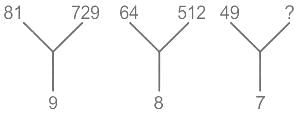

Detailed Solution: Question 4
When a child with a disability first comes to school the teacher should?
Detailed Solution: Question 5
Detailed Solution: Question 6
Detailed Solution: Question 7
One or More than One Options Correct Type
This section contains 5 multiple type questions. Each question has 4 choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Select the correct point(s) of distinction between a volatic cell and electrolysis cell.

Detailed Solution: Question 8
Detailed Solution: Question 9
Detailed Solution: Question 10
Electron-rich hydrides has excess electrons that are present as
Detailed Solution: Question 11
25.3 g of sodium carbonate, Na2CO3 is dissolved in enough water to make 250 mL of solution. If sodium carbonate dissociates completely, molar concentration of sodium ions, Na+ and carbonate ions, 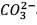 are respectively. (At. mass of carbon = 40)
are respectively. (At. mass of carbon = 40)
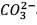 are respectively. (At. mass of carbon = 40)
are respectively. (At. mass of carbon = 40) Detailed Solution: Question 12
Consider the reactions
(A) H2O2 + 2HI → I2 +2H2O
(B) HOCl + H2O2 → H3O + Cl - - + O2
Which of the following statements is correct about H2O2 with reference to these reactions? Hydrogen perioxide is ________.
(B) HOCl + H2O2 → H3O + Cl - - + O2
Detailed Solution: Question 13
Consider the following equilibrium in a closed container
N2O4 (g)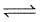 2NO2(g)
2NO2(g)
At a fixed temperature, the volume of a reaction container is halved. For this change, which of the following statements holds true regarding the equilibrium constant (Kp) and degree of dissociation (α) ?
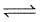 2NO2(g)
2NO2(g)Detailed Solution: Question 14
A definite amount of solid NH4HS is placed in a flask already containing ammonia gas at a certain temperature and 0.50 atm pressure. NH4HS decomposes to give NH3 and H2S and at equilibrium total pressure in flask is 0.84 atm. The equilibrium constant for the reaction is :
Detailed Solution: Question 15
If x denoted the number of valence electrons of a representative element then its valence is equal to:
Detailed Solution: Question 16
Detailed Solution: Question 17
In Dobereiner's Triads, elements were grouped based on their similar chemical properties. Which of the following elements was not part of any known Dobereiner's Triad?
Detailed Solution: Question 18
In an atom, an electron is moving with a speed of 600m/s with an accuracy of 0.005%. Certainity with which the position of the electron can be located is (h = 6.6 ×10−34 Js)
Detailed Solution: Question 19
Detailed Solution: Question 20
Which one of the following configuration represents a metallic character?
Detailed Solution: Question 21
A group of 14 element is converted into n – type semiconductor by dopping it with
Detailed Solution: Question 22
What are the roots of 1, 1, and 9 respectively as per the IUPAC nomenclature, and find out its symbol?
Detailed Solution: Question 23
What are the products in the following reaction?
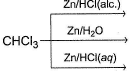
Detailed Solution: Question 24
Detailed Solution: Question 25
A coordination compound of cobalt has the molecular formula containing five ammonia molecules, one nitro group and two chlorine atoms for one cobalt atom. One mole of this compound produces three ions in an aqueous solution. The aqueous solution on treatment with an excess of AgNO3 gives two moles of AgCI as a precipitate. The formula of the complex and the isomerism shown by this is
Detailed Solution: Question 26
Examine the compound on the right. How many stereoisomers having this constitution are possible ?
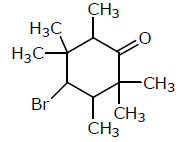
Detailed Solution: Question 27
Number of neighbours and next - nearest neighbours of K respectively, are
Detailed Solution: Question 28
In Calgon’s method, one of the following chemical is used to remove hardness of water
Detailed Solution: Question 29
Pick out the pair of species having identical shapes for both the molecules.
Detailed Solution: Question 30



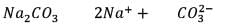
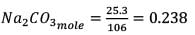
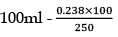
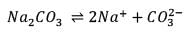
 0.955
0.955 