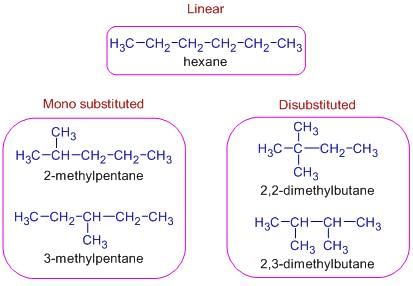
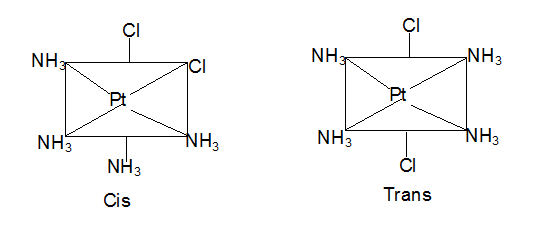
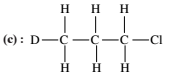
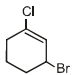
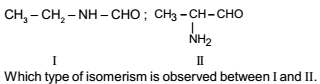
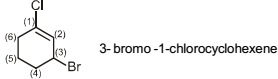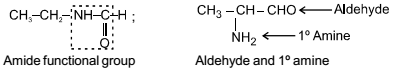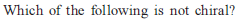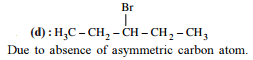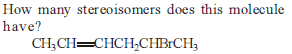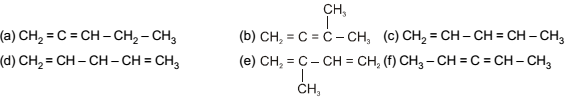Isomerism - 1 - Free MCQ Test with solutions for JEE Chemistry
MCQ Practice Test & Solutions: Isomerism - 1 (30 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Isomerism - 1". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
Detailed Solution: Question 2
Detailed Solution: Question 3
Detailed Solution: Question 4
What is the specific rotation if its observed rotation is given as 3x, its length is given as x and density is given as 3/y?
Detailed Solution: Question 5
Detailed Solution: Question 6
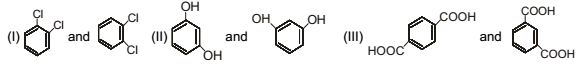
Detailed Solution: Question 7
Detailed Solution: Question 8
Detailed Solution: Question 9
Detailed Solution: Question 10
Detailed Solution: Question 11
Detailed Solution: Question 12
Detailed Solution: Question 13
Detailed Solution: Question 14
Detailed Solution: Question 15
Detailed Solution: Question 16
Detailed Solution: Question 17
How many number of all structurally isomeric dienes with molecular formula C5H8 are possible :
Detailed Solution: Question 18
S1 : Trans-But-2-ene has higher boiling point than cis-But-2-ene.
S2 : 1, 4-Dichlorobenzene has zero dipole moment.
S3 : Trans cyclodecene is more stable as compare to cis-cyclodecene.
S4 : Trans 1, 2-Dibromoethene is more soluble in water than cis-1, 2-Dibromoethene
S2 : 1, 4-Dichlorobenzene has zero dipole moment.
S3 : Trans cyclodecene is more stable as compare to cis-cyclodecene.
S4 : Trans 1, 2-Dibromoethene is more soluble in water than cis-1, 2-Dibromoethene
Detailed Solution: Question 19
Which statement(s) is/are true about the relation between the following compounds?
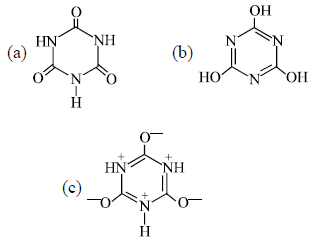
(I) a and b are tautomers
(II) b and c are resonating structures
(III) a and c are resonating structures
(IV) a and c are tautomers

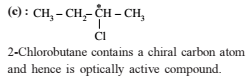
Which of the following compound has zero dipole moment in one of the stable conformations
Detailed Solution: Question 21
The number of structurally isomeric esters with molecular formula C5H10O2 are.
Detailed Solution: Question 22
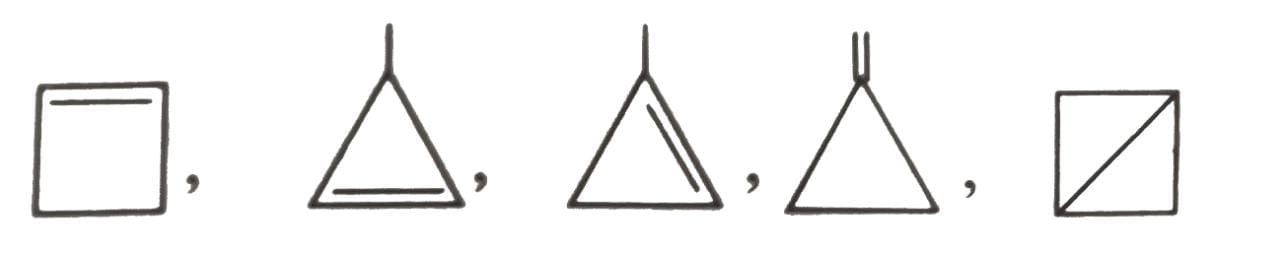
The total number of cyclic isomers possible for a hydrocarbon with the molecular formula C4H6 is / are :
Detailed Solution: Question 23
Detailed Solution: Question 24
What is the degree of unsaturation in a compound with molecular formula C9H6N4?
Detailed Solution: Question 25
Detailed Solution: Question 26
Detailed Solution: Question 27
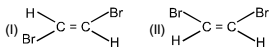
Which of the following physical property is greater for (I) compound.
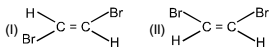
Detailed Solution: Question 28
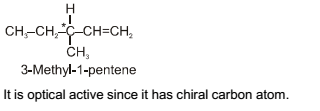
Out of the following, the alkene that exhibits optical isomerism is.
Detailed Solution: Question 29
Detailed Solution: Question 30
335 videos|699 docs|300 tests |