JEE Advanced Practice Test- 7 National Level Test Series MCQs & solutions
MCQ Practice Test & Solutions: JEE Advanced Practice Test- 7 (54 Questions)
You can prepare effectively for JEE National Level Test Series for JEE Advanced 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "JEE Advanced Practice Test- 7". These 54 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 180 minutes
- - Number of Questions: 54
Sign up on EduRev for free to attempt this test and track your preparation progress.
A tank of uniform cross-section is completely filled with ice. The height of ice is H and its mass is m. When the entire ice melts, the work done by gravity is
(ρice = 0.9 gm/cc, ρwater = 1 g/cc and g represents acceleration due to gravity)
Detailed Solution: Question 1
A hydrogen like species having atomic number Z = 2, in ground state, is excited by means of electromagnetic radiation of frequency 1.315 × 1016 Hz. How many spectral lines will be observed in the emission spectrum?
(Planck’s constant h = 4.14 × 10-15 eVs)
Detailed Solution: Question 2
Velocity of a point on the equator of a rotating spherical planet is v. The angular velocity of the planet is such that the apparent value of acceleration due to gravity ‘g’ at the equator is half of that at the poles. The escape velocity of a particle from the surface of the planet is
Detailed Solution: Question 3
A positive charge q is projected from origin with a velocity along positive x-axis in a region having uniform magnetic field directed towards negative y-axis. If T is the time period of circular motion then the velocity vector of charge q at some instant t where 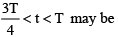
Detailed Solution: Question 4
Four charges q1, q2, q3 and q4 are placed at the positions as shown in the figure, given q1 +q2+ q3 + q4 = 0 . The electric field on z-axis
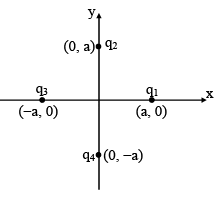
Detailed Solution: Question 5
For the circuit as shown in the figure q1 and q2 be the charges on 3μF and 6μF capacitors respectively, then
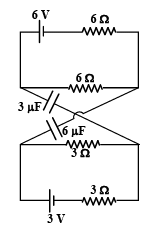
Detailed Solution: Question 6
A ball is released from position A and drops 10 m before striking a smooth incline. The coefficient of 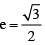 . If the time taken by the ball to strike the incline again is t then find the value of t2 [in (second)2]. (g = 10 m/s2)
. If the time taken by the ball to strike the incline again is t then find the value of t2 [in (second)2]. (g = 10 m/s2)
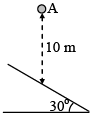
Detailed Solution: Question 7
Two identical beads P and Q of mass 1 kg each are connected by an inextensible massless string and they can slide along the two arms AB and BC of a rigid smooth wire frame in vertical plane. If the system is released from rest and vQ is the speed of bead Q when they have both moved by a distance of 0.1 m then find the value of 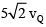 (in m/s). (g = 10 m/s2)
(in m/s). (g = 10 m/s2)
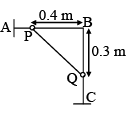
Detailed Solution: Question 8
A solid uniform sphere rotating about its axis with kinetic energy Eo is gently placed on a rough horizontal plane. The coefficient of friction on the plane varies from point to point. After some time, the sphere begins pure rolling with total kinetic energy equal to E. Then find the value of 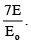
Detailed Solution: Question 9
A variable voltage V = 2t is applied across an inductor of inductance L = 2H as shown in figure. Then find the rate at which magnetic potential energy stored in the inductor is increasing at t = 1 s (in J/s). Take the current through the inductor at t = 0 as zero.
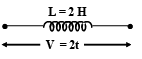
Detailed Solution: Question 10
A calorimeter of mass m contains an equal mass of water in it. The temperature of water and calorimeter is t2. A block of ice of mass m and temperature t3 < 0oC is gently dropped into the calorimeter. Let C1, C2 and C3 be the specific heats of calorimeter, water and ice respectively and L be the latent heat of fusion of ice.
Q.
The whole mixture in the calorimeter becomes ice if
Detailed Solution: Question 11
A calorimeter of mass m contains an equal mass of water in it. The temperature of water and calorimeter is t2. A block of ice of mass m and temperature t3 < 0oC is gently dropped into the calorimeter. Let C1, C2 and C3 be the specific heats of calorimeter, water and ice respectively and L be the latent heat of fusion of ice.
Q.
The whole mixture in the calorimeter becomes water if
Detailed Solution: Question 12
A calorimeter of mass m contains an equal mass of water in it. The temperature of water and calorimeter is t2. A block of ice of mass m and temperature t3 < 0oC is gently dropped into the calorimeter. Let C1, C2 and C3 be the specific heats of calorimeter, water and ice respectively and L be the latent heat of fusion of ice.
Q.
Water equivalent of calorimeter is
Detailed Solution: Question 13
A parallel plate capacitor has its plate horizontal with air occupying the space between the plates. The upper plate is fixed with a rigid support and the lower one is connected to a spring as shown. The distance between the plates is d1. Now the capacitor is connected with an electric source having voltage V. The separation between the plates changes to d2 at equilibrium. The mass of lower plate is ‘m’ and cross-sectional area of each plate is A.
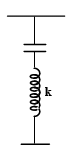
Q.
The spring constant k is
Detailed Solution: Question 14
A parallel plate capacitor has its plate horizontal with air occupying the space between the plates. The upper plate is fixed with a rigid support and the lower one is connected to a spring as shown. The distance between the plates is d1. Now the capacitor is connected with an electric source having voltage V. The separation between the plates changes to d2 at equilibrium. The mass of lower plate is ‘m’ and cross-sectional area of each plate is A.

Q.
The maximum voltage Vm for a given k for which an equilibrium exists is
Detailed Solution: Question 15
A parallel plate capacitor has its plate horizontal with air occupying the space between the plates. The upper plate is fixed with a rigid support and the lower one is connected to a spring as shown. The distance between the plates is d1. Now the capacitor is connected with an electric source having voltage V. The separation between the plates changes to d2 at equilibrium. The mass of lower plate is ‘m’ and cross-sectional area of each plate is A.

Q.
When lower plate is slightly displaced about equilibrium position, time period T of small oscillations is
Detailed Solution: Question 16
A certain weak acid has a dissociation constant 1.0 x 10-4. The equilibrium constant for its reaction with strong base is
Detailed Solution: Question 17
The major product formed in the following reaction is
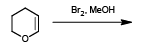
Detailed Solution: Question 18
Identify the major product of the following reaction:
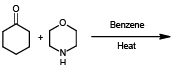
Detailed Solution: Question 19
The oxidation state of molybdenum in [(η7-tropylium)Mo(CO)3]+ is
Detailed Solution: Question 20
In metal-olefin interaction, the extent of increase in metal ⎯→ olefin π-back-donation would
The highest oxidation state of an element in the following compound that behaves as an acid in H2SO4 is
AcOH, HNO2, HNO3, H2O, HClO, HClO4
Detailed Solution: Question 22
How many unpaired electrons are present in O2 molecule?
One mole of Pb3O4 is separately reacted with excess of HCl and HNO3. The difference in moles of HCl and HNO3 is
Detailed Solution: Question 24
How many moles of phenyl hydrazine are used in the formation of osazone from glucose?
Detailed Solution: Question 25
Detailed Solution: Question 26
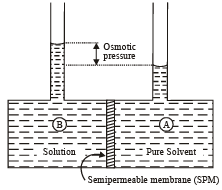
If a concentrated solution of copper sulphate is placed at the bottom of a beaker of water or that of a dilute solution of copper sulphate is carefully poured over it there will be a two distinct layer visible. However, after some time the boundaries will disappear. This property is called diffusion. If we now consider a solution which is separated from the pure solvent by a semipermeable membrane then the solvent particles move from the pure solvent region through the SPM to the solution region. This phenomenon is called osmosis.

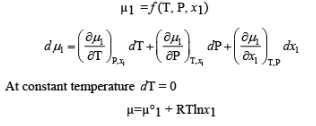
Q.
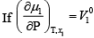 for an indefinitely dilute boundary then dμ1 is
for an indefinitely dilute boundary then dμ1 is
If a concentrated solution of copper sulphate is placed at the bottom of a beaker of water or that of a dilute solution of copper sulphate is carefully poured over it there will be a two distinct layer visible. However, after some time the boundaries will disappear. This property is called diffusion. If we now consider a solution which is separated from the pure solvent by a semipermeable membrane then the solvent particles move from the pure solvent region through the SPM to the solution region. This phenomenon is called osmosis.

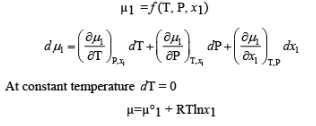

Q.
Which of the following solution has highest osmotic pressure?
If a concentrated solution of copper sulphate is placed at the bottom of a beaker of water or that of a dilute solution of copper sulphate is carefully poured over it there will be a two distinct layer visible. However, after some time the boundaries will disappear. This property is called diffusion. If we now consider a solution which is separated from the pure solvent by a semipermeable membrane then the solvent particles move from the pure solvent region through the SPM to the solution region. This phenomenon is called osmosis.

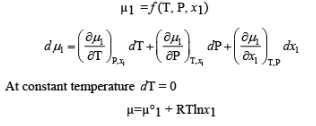

Q.
Which of the following is correct?
Thionyl chloride can be synthesized by chlorinating SO2 using PCl5. Thionyl chloride is used to prepare anhydrous ferric chloride starting from its hexahydrated salt. Alternatively, the anhydrous ferric chloride can also be prepared from hexahydrated salt by treating with 2,2-dimethoxypropane.
Q.
Consider the following reaction
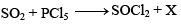
The compound X is
The compound X is
Detailed Solution: Question 30
1 videos|14 docs|24 tests |


