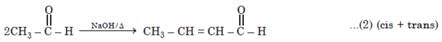JEE Main Chemistry Mock Test- 1 Free Online Test 2026
Full Mock Test & Solutions: JEE Main Chemistry Mock Test- 1 (25 Questions)
You can boost your JEE 2026 exam preparation with this JEE Main Chemistry Mock Test- 1 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of JEE 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Total Questions: 25
- - Analysis: Detailed Solutions & Performance Insights
- - Sections covered: Section I, Section II
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
Detailed Solution: Question 1
Detailed Solution: Question 2
Detailed Solution: Question 3
Chemical equilibrium is dynamic in nature, because
Detailed Solution: Question 4
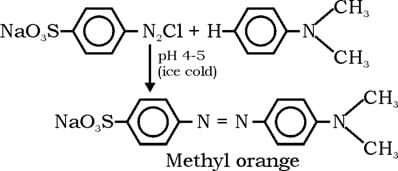
The indicator that is obtained by coupling the diazonium salt of sulfanilic acid with N,N-dimethylaniline is
Detailed Solution: Question 5
The standard e.m.f. of a galvanic cell, with standard electrode potentials of zinc equal to -0.76 V and that of copper equal to + 0.34 V, is
Detailed Solution: Question 6
Which of the following is not true for the order of a reaction?
Detailed Solution: Question 7
When 10gram of methane is completely burnt in oxygen, the heat evolved is 560kJ. What is the heat of combustion (in kJ/mole) of methane?
Detailed Solution: Question 8
Detailed Solution: Question 9
The catalyst used for the polymerization of olefins is
Detailed Solution: Question 10
When one Faraday current is passed which of the following would deposit one gram atomic weight of the metal?
Detailed Solution: Question 11
A white salt is readily solubule in water and gives a colourless solution with a pH of about 9. The salt would be
Detailed Solution: Question 12
The pH of a solution of hydrochloric acid is 4. The molarity of this solution is
Detailed Solution: Question 13
Which one of the following can be considered as weak electrolyte ?
Detailed Solution: Question 14
Detailed Solution: Question 15
Detailed Solution: Question 16
Detailed Solution: Question 17
A solid cube has CsCl type structure. If the edge length of the unit cell is 404 pm, what is the distance between A⁺ and B⁻ ions?
Detailed Solution: Question 18
Which of the following has maximum Schottky defect?
Detailed Solution: Question 19
Select the correct statement regarding the aromatic nitrogen molecule
Detailed Solution: Question 20
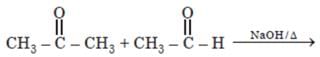
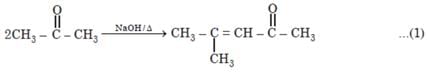
Number of possible self aldol condensation product on heating with NaOH?
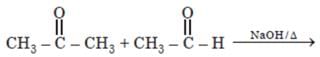
Number of possible self aldol condensation product on heating with NaOH?
Detailed Solution: Question 21
Among the following the total number of elements which produce H2 gas with NaOH is Zn, Al, Sn, Pb, P, S
Detailed Solution: Question 22
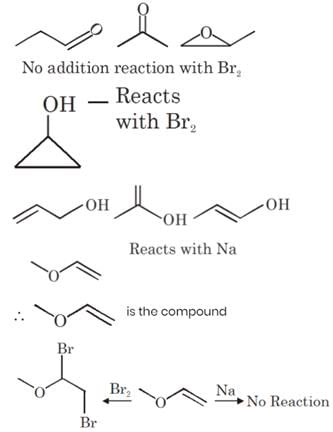
An organic compound P contains 62.07% carbon and 10.34% hydrogen and rest oxygen. Its vapour density is 29. This compound does not react with sodium metal, but its 2.9 g combines with X g of bromine (to give dibromo addition product). Find out value of (Y–X). (Where Y is total number of possible isomers of given organic compound P)
[Atomic mass : Br = 80]
[Atomic mass : Br = 80]
Detailed Solution: Question 23
How many of the following reagent/s will liberate at least one oxide of nitrogen as a product ?
(i) Ag + conc. HNO3
(ii) Sn + dil., HNO3 (20%)
(iii) Cu + conc. HNO3
(iv) C + conc. HNO3
(v) Zn + conc, HNO3
(vi) Zn + dil. HNO3 (20%)
(vii) P4 + conc. HNO3
(viii) S8 + conc. HNO3
(ix) Cu + dil. HNO3 (20%)
(i) Ag + conc. HNO3
(ii) Sn + dil., HNO3 (20%)
(iii) Cu + conc. HNO3
(iv) C + conc. HNO3
(v) Zn + conc, HNO3
(vi) Zn + dil. HNO3 (20%)
(vii) P4 + conc. HNO3
(viii) S8 + conc. HNO3
(ix) Cu + dil. HNO3 (20%)
Detailed Solution: Question 24
In white phosphorous (P4) if
x is total number of triangle
y is total number of plane of symmetry z is total number of P–P bond
then calculate value of (y + z)/x.
x is total number of triangle
y is total number of plane of symmetry z is total number of P–P bond
then calculate value of (y + z)/x.
Detailed Solution: Question 25
360 docs|100 tests |