JEE Main Chemistry Mock Test- 3 Free Online Test 2026
Full Mock Test & Solutions: JEE Main Chemistry Mock Test- 3 (25 Questions)
You can boost your JEE 2026 exam preparation with this JEE Main Chemistry Mock Test- 3 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of JEE 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Total Questions: 25
- - Analysis: Detailed Solutions & Performance Insights
- - Sections covered: Section I, Section II
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
The compound 'A' when treated with methanol and few drops of H₂SO₄ give wintergreen smell. The compound 'A' is
Detailed Solution: Question 1
1-Phenylethanol can be prepared by the reaction of benzaldehyde with
Detailed Solution: Question 2
The spectrum produced due to transition of an electron from M to L shell is
Detailed Solution: Question 3
The correct order of increasing energy of atomic orbitals is
Detailed Solution: Question 4
Detailed Solution: Question 5
Elimination of bromine from 2- bromobutane results in the formation of
Detailed Solution: Question 6
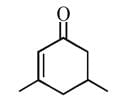
The name of 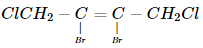 according to IUPAC nomenclature system is
according to IUPAC nomenclature system is
Detailed Solution: Question 7
Detailed Solution: Question 8
The bond order in NO is 2.5 while that in NO+ is 3. Which of the following statements is true for these two species?
Detailed Solution: Question 9
The segment of DNA which acts as the instructional manual for the synthesis of the protein is
Detailed Solution: Question 10
Detailed Solution: Question 11
What are the products formed in the reaction of xenon hexafluoride with silicon dioxide
Detailed Solution: Question 12
The hardness of transition elements is due to their
Detailed Solution: Question 13
Sodium nitroprusside reacts with sulphide ion to give a purple colour due to the formation of :
Detailed Solution: Question 14
To a solution containing equimolar mixture of sodium acetate and acetic acid, some more amount of sodium acetate solution is added. The pH of mixture solution
Detailed Solution: Question 15
The reason for geometrical isomerism by 2-butene is
Detailed Solution: Question 16
Detailed Solution: Question 17
If chloroform is left open in air in presence of sunlight
Detailed Solution: Question 18
The bond dissociation energy of B–F in BF3 is 646 kJ mol-1 whereas that of C–F in CF4 is 515 kJ mol–1. The correct reason for higher B–F bond dissociation energy as compared to that of C–F is
Detailed Solution: Question 19
At 35°C, the vapor pressure of CS2 is 512 mm Hg and that of acetone is 344 mm Hg. A solution of CS2 in acetone has a total vapor pressure of 600 mm Hg. The false statement amongst the following is (2020)
(a) Raoult’s law is not obeyed by this system.
(b) A mixture of 100 mL CS2 and 100 mL acetone has a volume < 200 mL.
(c) CS2 and acetone are less attracted to each other than to themselves.
(d) Heat must be absorbed in order to produce the solution at 35°C.
(a) Raoult’s law is not obeyed by this system.
(b) A mixture of 100 mL CS2 and 100 mL acetone has a volume < 200 mL.
(c) CS2 and acetone are less attracted to each other than to themselves.
(d) Heat must be absorbed in order to produce the solution at 35°C.
Detailed Solution: Question 20
A vessel at 1000 K contains CO2 with a pressure of 0.5 atm. Some of the CO2 is converted into CO on the addition of graphite. If the total pressure at equilibrium is 0.8 atm, the value of K is
Detailed Solution: Question 21
If x = Group number of Europium (atomic number 63)
y = Period number of Americium (atomic number 95)
z = Most stable oxidation state shown by Gadolinium (atomic number 64)
then find the value of x + y + z?
y = Period number of Americium (atomic number 95)
z = Most stable oxidation state shown by Gadolinium (atomic number 64)
then find the value of x + y + z?
Detailed Solution: Question 22
The decomposition of hydrogen peroxide follows first order kinetics.
2H2O2(aq) → 2H2O(ℓ) + O2(g)
If the volume of O2 gas liberated at STP in first 20 minutes of the start of decomposition is 25.00 ml, what should be the total volume of O2 gas (in ml) collected in time, t >> t1/2.
(t1/2 for the decomposition of H2O2 is 10 min)
2H2O2(aq) → 2H2O(ℓ) + O2(g)
If the volume of O2 gas liberated at STP in first 20 minutes of the start of decomposition is 25.00 ml, what should be the total volume of O2 gas (in ml) collected in time, t >> t1/2.
(t1/2 for the decomposition of H2O2 is 10 min)
Detailed Solution: Question 23
How many H-atom can be exchanged by D-atom when the following compound is kept in 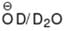 solution for long time?
solution for long time?
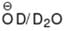 solution for long time?
solution for long time?
Detailed Solution: Question 24
Find out the number of following orders which are INCORRECT against the mentioned properties :
(i) Ortho hydrogen > Para hydrogen (Stability at low temperature)
(ii) Ethanol > Glycerol (Viscosity)
(iii) D2 < He (Boiling Point)
(iv) HF > H2O (Melting point)
(v) H3BO3 > BF3 (Melting point)
(vi) NH3 < SbH3 (Boiling point)
(vii) H2O2 > H2O (Strength of hydrogen bond)
(viii) H2O < D2O (Freezing point)
(ix) 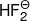 < HF (Strength of hydrogen bond)
< HF (Strength of hydrogen bond)
(x) D2O < H2O (Bond energy)
(i) Ortho hydrogen > Para hydrogen (Stability at low temperature)
(ii) Ethanol > Glycerol (Viscosity)
(iii) D2 < He (Boiling Point)
(iv) HF > H2O (Melting point)
(v) H3BO3 > BF3 (Melting point)
(vi) NH3 < SbH3 (Boiling point)
(vii) H2O2 > H2O (Strength of hydrogen bond)
(viii) H2O < D2O (Freezing point)
(ix)
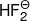 < HF (Strength of hydrogen bond)
< HF (Strength of hydrogen bond)(x) D2O < H2O (Bond energy)
Detailed Solution: Question 25
360 docs|100 tests |