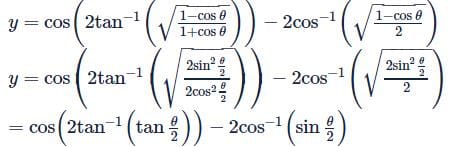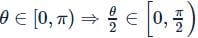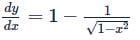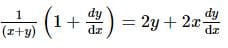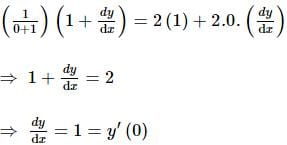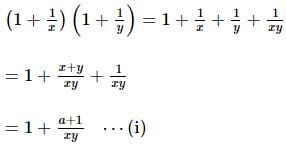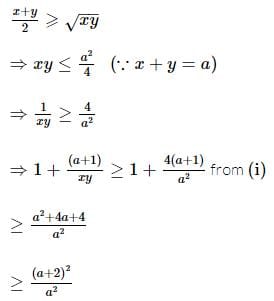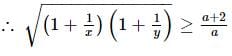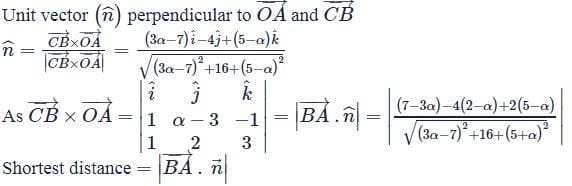JEE Main Mock Test - 10 Free Online Test 2026
Full Mock Test & Solutions: JEE Main Mock Test - 10 (75 Questions)
You can boost your JEE 2026 exam preparation with this JEE Main Mock Test - 10 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of JEE 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 180 minutes
- - Total Questions: 75
- - Analysis: Detailed Solutions & Performance Insights
- - Sections covered: Physics: Section A, Physics: Section B, Chemistry: Section A, Chemistry: Section B, Mathematics: Section A, Mathematics: Section B
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
In the following a statement of Assertion is followed by a statement of Reason.
Assertion: The mass defect involved in a chemical reaction is almost a million times smaller than that in a nuclear reaction.
Reason: The mass energy interconversion does not take place in a chemical reaction.
Assertion: The mass defect involved in a chemical reaction is almost a million times smaller than that in a nuclear reaction.
Reason: The mass energy interconversion does not take place in a chemical reaction.
Detailed Solution: Question 1
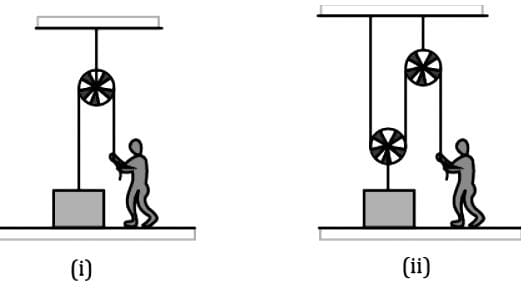
In the figure shown, a person wants to raise a block lying on the ground to a height h. In both the cases if time required is same then in which case he has to exert more force. Assume pulleys and strings light.

Detailed Solution: Question 2
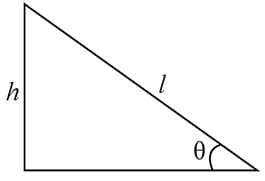
A railway track is banked for a speed v, by making the height of the outer rail h higher than that of the inner rail. If the distance between the rails is l and the radius of curvature of the track is r, then
Detailed Solution: Question 3
In vernier callipers instrument 20 vernier scale divisions concide with 18 main scale divisions where 1 mm=1 main scale division. The least count is
Detailed Solution: Question 4
If the wavelength of the first line of the Balmer series of hydrogen is 6561Å, find the wavelength of the second line of the series.
Detailed Solution: Question 5
Two identical conducting spheres, A and B, have equal charges and are separated at a distance such that charge on each of them is essentially uniform on its surface. Athird identical conducting neutral sphere C, is brought in contact with A and then in contact with B and removed far away. If initial force between the two spheres A and B was F, then the new force between them would be (Assuming that in calculating both the forces, distance of separation remains same) 3F/P, Find integral value of P.
Detailed Solution: Question 6
A long straight wire of radius a carries a steady current i. The current is uniformly distributed across its cross section. The ratio of the magnetic field at a/2 and 2a is,
Detailed Solution: Question 7
The magnetic flux through a circuit of resistance Rchanges by an amount Δϕ in a time Δt. The total magnitude of electric charge Q, that passes through any point in the circuit during this time is
Detailed Solution: Question 8
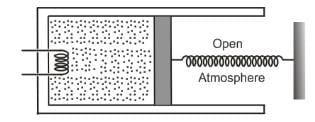
An ideal monatomic gas is confined in a cylinder by a spring-loaded, massless piston with a cross-sectional area of 8×10⁻³ m². The piston moves frictionlessly inside the cylinder. Initially, the gas is at 300 K and occupies a volume of 2.4×10⁻³ m³, with the spring in its relaxed position. The gas is then heated slowly using an electric heater, causing the piston to move out by 0.1 m.
Find the final temperature of the gas and the heat supplied by the heater.
Given:
Force constant of the spring, k = 8000 N/m
Atmospheric pressure, P₀ = 1×10⁵ N/m²
Find the final temperature of the gas and the heat supplied by the heater.
Given:
Force constant of the spring, k = 8000 N/m
Atmospheric pressure, P₀ = 1×10⁵ N/m²
Detailed Solution: Question 9
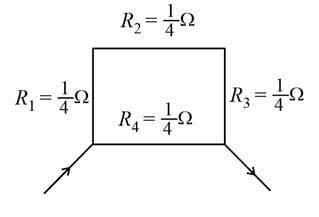
The resistance of the rod is 1 Ω. It is bent in form of a square. What is the resistance across adjacent corners?
Detailed Solution: Question 10
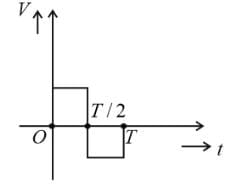
The current (I) in the inductance is varying with time according to the plot shown in the figure.
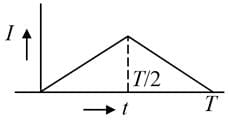
The correct variation of voltage with time in the coil is

Detailed Solution: Question 11
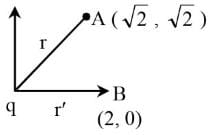
An electric charge 10–3 μC is placed at origin (0, 0). Two points A and B are situated at (√2¸ √2) and (2, 0) respectively. The potential difference between the points A and B will be(in J):
Detailed Solution: Question 12
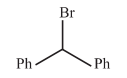
The rate SN1 reaction will be faster for which of the following bromides?
Detailed Solution: Question 13
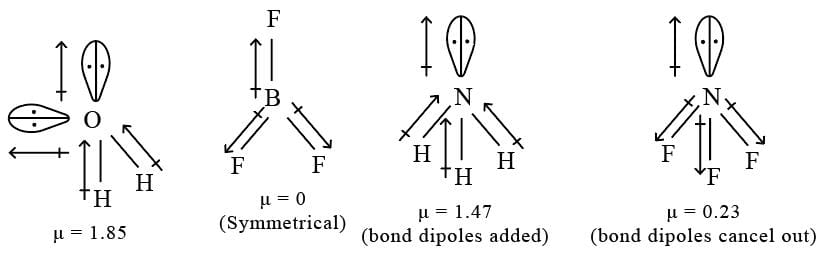
Assertion : First ionization energy for nitrogen is lower than oxygen.
Reason : Across a period effective nuclear charge decreases.
Reason : Across a period effective nuclear charge decreases.
Detailed Solution: Question 14
Detailed Solution: Question 15
In an atom, the order of increasing energy of electrons with quantum numbers:
(i) n = 4, ℓ = 1
(ii) n = 4, ℓ = 0
(iii) n = 3, ℓ = 2
(iv) n = 3, ℓ = 1
The correct order is:
(i) n = 4, ℓ = 1
(ii) n = 4, ℓ = 0
(iii) n = 3, ℓ = 2
(iv) n = 3, ℓ = 1
The correct order is:
Detailed Solution: Question 16
The order of strengths of the following carboxylic acids is
(i) CH3−CH2−COOH
(ii) CH3−COOH
(iii) C6H5−COOH
(iv) C6H5−CH2COOH
(i) CH3−CH2−COOH
(ii) CH3−COOH
(iii) C6H5−COOH
(iv) C6H5−CH2COOH
Detailed Solution: Question 17
The correct option(s) to distinguish nitrate salts of Mn2+ and Cu2+ taken separately is (are)
(1) Mn2+ shows the characteristic green colour in the flame test
(2) only Cu2+ shows the formation of precipitate by passing H2 S in acidic medium
(3) only Mn2+ shows the formation of precipitate by passing H2 S in faintly basic medium
(4) Cu2+∣Cu has higher reduction potential than Mn2+ ∣Mn (measured under similar conditions)
(1) Mn2+ shows the characteristic green colour in the flame test
(2) only Cu2+ shows the formation of precipitate by passing H2 S in acidic medium
(3) only Mn2+ shows the formation of precipitate by passing H2 S in faintly basic medium
(4) Cu2+∣Cu has higher reduction potential than Mn2+ ∣Mn (measured under similar conditions)
Detailed Solution: Question 18
Detailed Solution: Question 19
Which of the following cannot be synthesized by Stephen's reaction ?
Detailed Solution: Question 20
How much NaNO₃ must be weighed out to make 50 mL of an aqueous solution containing 70 mg of Na⁺ per mL?
Detailed Solution: Question 21
Consider the following reversible reaction:
NO + NO3 ⇌ 2NO2
If 1.0mol of NO is mixed with 3.0mol of NO3 , 'x' mole of NO2 is produced at equilibrium. If 2.0mol of NO is added further, 'x' mol of NO2 is further produced. What is the value of equilibrium constant?
NO + NO3 ⇌ 2NO2
If 1.0mol of NO is mixed with 3.0mol of NO3 , 'x' mole of NO2 is produced at equilibrium. If 2.0mol of NO is added further, 'x' mol of NO2 is further produced. What is the value of equilibrium constant?
Detailed Solution: Question 22
The coordinates of the perpendicular drawn from the point 2î - ĵ + 5k̅ to the line r̅ = (11î - 2ĵ - 8k̅) + λ(10î - 4ĵ - 11k̅) are: (1, 2, 3).
Detailed Solution: Question 23
Let f(x) be defined by 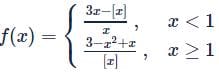 (where [.] is GIF) Then which is incorrect
(where [.] is GIF) Then which is incorrect
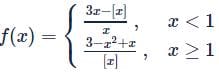 (where [.] is GIF) Then which is incorrect
(where [.] is GIF) Then which is incorrectDetailed Solution: Question 24
Detailed Solution: Question 25
The range of the function y = 2sin−1[x2 + 1/2] + cos−1[x2 − 1/2] is (where, [⋅] denotes the greatest integer function)
Detailed Solution: Question 26
Detailed Solution: Question 27
If x and y are two positive numbers such that x + y = a, then the minimum value of 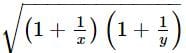 is
is
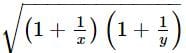 is
isDetailed Solution: Question 28
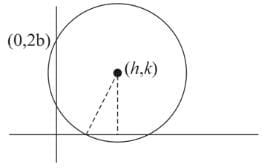
A circle cuts a chord of length 4a on the line y = 0 and passes through a point on the line x = 0, having distance 2b from the origin, then the locus of the centre of this circle is:
Detailed Solution: Question 29
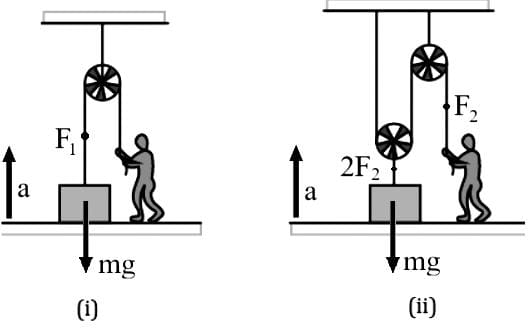
OABC is a tetrahedron with O as the origin and position vectors of points A, B, C as:
- A → î + 2ĵ + 3k̂
- B → 2î + αĵ + k̂
- C → î + 3ĵ + 2k̂
If the shortest distance between 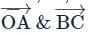 is √(3/2), then the integral value of α is ___.
is √(3/2), then the integral value of α is ___.
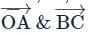 is √(3/2), then the integral value of α is ___.
is √(3/2), then the integral value of α is ___.Detailed Solution: Question 30
360 docs|100 tests |



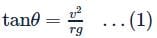
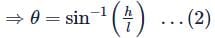
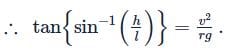
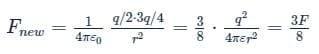
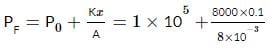
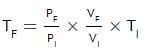
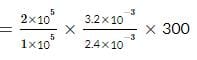

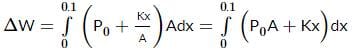
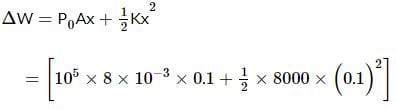
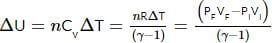
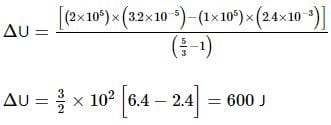
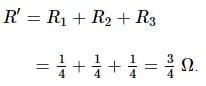
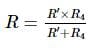
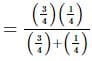
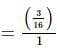
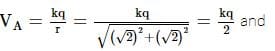
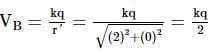
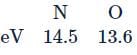
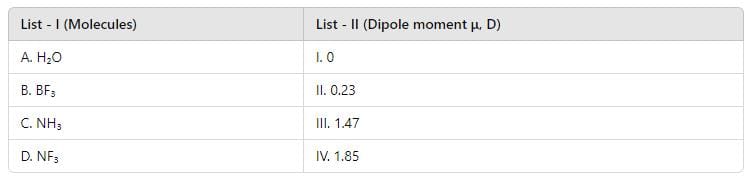
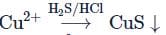
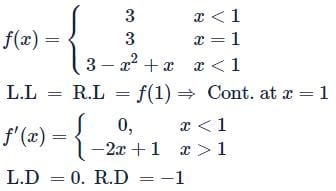
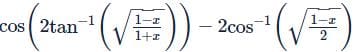 W. r. to x is
W. r. to x is