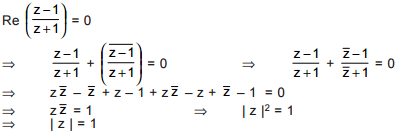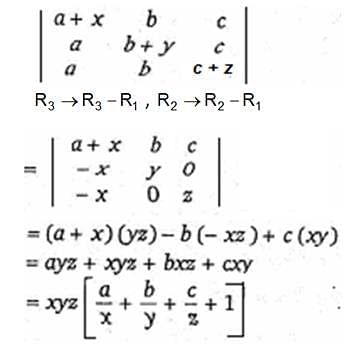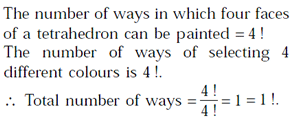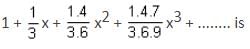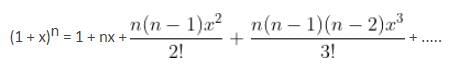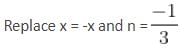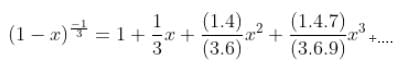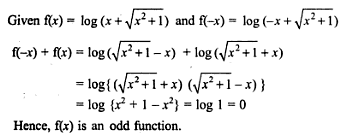KIITEE Mock Test - 1 Free Online Test 2026
Full Mock Test & Solutions: KIITEE Mock Test - 1 (120 Questions)
You can boost your JEE 2026 exam preparation with this KIITEE Mock Test - 1 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of JEE 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 180 minutes
- - Total Questions: 120
- - Analysis: Detailed Solutions & Performance Insights
- - Sections covered: Physics, Chemistry, Maths
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
The magnetic field in a region is given by 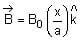 . A square loop of side d is placed with its edges along the x and y-axis. The loop is moved with a constant velocity
. A square loop of side d is placed with its edges along the x and y-axis. The loop is moved with a constant velocity 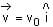 . The emf induced in the loop is
. The emf induced in the loop is
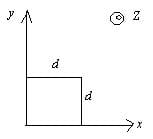
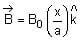 . A square loop of side d is placed with its edges along the x and y-axis. The loop is moved with a constant velocity
. A square loop of side d is placed with its edges along the x and y-axis. The loop is moved with a constant velocity 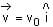 . The emf induced in the loop is
. The emf induced in the loop is
Detailed Solution: Question 1
The output current (I) versus time (t) curve of a rectifier is shown in the figure. The average value of the output current in this case is
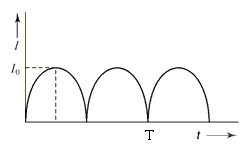

Detailed Solution: Question 2
The angle of minimum deviation for a prism of refractive index 3/2 is equal to the angle of prism. The angle of prism is
Detailed Solution: Question 3
Detailed Solution: Question 4
A moving coil galvanometer consists of a coil of N turns and area A suspended by a thin phosphor bronze strip in radial magnetic field B. The moment of inertia of the coil about the axis of rotation is l and C is the torsional constant of the phosphor bronze strip. When a current i is passed through the coil, it deflects through an angle θ (in radians). The current sensitivity of the galvanometer is increased if
Detailed Solution: Question 5
A wire of length L metres carrying a current I amperes is bent in the form of a circle. The magnitude of the magnetic moment is
Detailed Solution: Question 6
In a single-slit diffraction experiment, the width of the slit is made double its original width. The central maximum of the diffraction pattern will become
Detailed Solution: Question 7
Two diodes have a resistance of 20 ohm and are centre-tapped with rms secondary voltage from centre tap to each end of secondary voltage of 50 V. If the external resistance is 980 ohm, then what is the mean load?
Detailed Solution: Question 8
If the potential energy of electron in a hydrogen atom is -Ke2/r, then its kinetic energy is
Detailed Solution: Question 9
When a thin transparent plate of thickness t and refractive index m is placed in the path of one of the two interfering waves of light, then the path difference changes by
Detailed Solution: Question 10
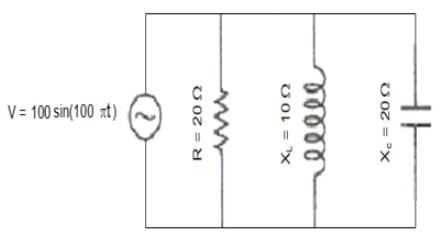
The voltage 7.25 sin (300t) is applied to a series RLC circuit with R = 120 ohms, L = 0.140 H and C = 1.45 μF. What is the impedance Z and the phase angle θ?
Detailed Solution: Question 11
A moving coil galvanometer consists of a coil of N turns and area A, suspended by a thin phosphor bronze strip in radial magnetic field B. The moment of inertia of the coil about the axis of rotation is l and C is the torsional constant of the phosphor bronze strip. When a current i is passed through the coil, it deflects through an angle θ (in radians).
When a charge Q is passed almost instantly through the coil, the angular speed ω acquired by the coil is
When a charge Q is passed almost instantly through the coil, the angular speed ω acquired by the coil is
Detailed Solution: Question 12
Detailed Solution: Question 13
By ignoring Aufbau principle, if each orbital accommodates three electrons instead of two, then the incorrect statement(s) about the new electronic arrangement of Zr atom having atomic number 40 is/are:
Detailed Solution: Question 14
Solubility product of a salt AB is 1 x 10-8 M2 in a solution in which the concentration of A+ ions is 10-3 M. The salt will precipitate when the concentration of B- ions is kept
Detailed Solution: Question 15
The compound [Co(H2O)6]2+, having three unpaired electrons, shows the experimental magnetic moment of 4.40 BM, while the theoretical calculated value is 3.87 BM. This is because of
Detailed Solution: Question 16
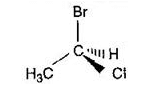
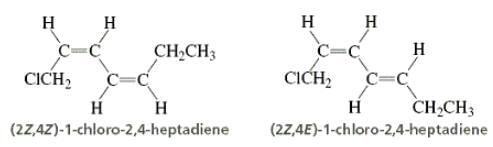
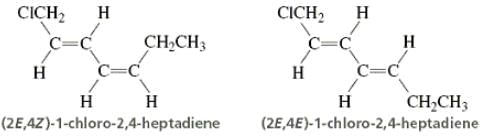
Directions: Consider the IUPAC names.
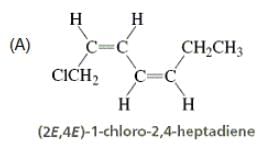
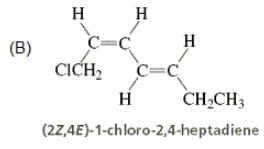
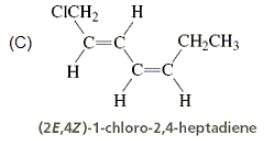
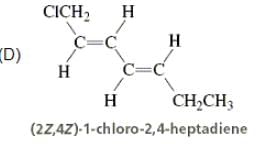
The incorrectly matched pairs are




Detailed Solution: Question 17
If 10-4 dm3 of water is introduced into a 1.0 dm3 flask at 300 K, then how many moles of water are in the vapour phase when the equilibrium is established?
(Given: Vapour pressure of H2O at 300 K is 3170 Pa and R = 8.314 J K-1 mol.)
Detailed Solution: Question 18
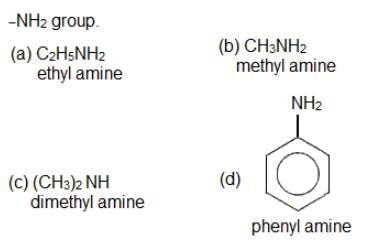
Which of the following amines will not undergo carbylamine reaction?
Detailed Solution: Question 19
During acetylation of amines, what is replaced by acetyl group?
Detailed Solution: Question 20
A hydrocarbon contains 10.5 g of carbon per g of H. If one litre vapour of hydrocarbon at 127°C and 1 atm pressure weighs 2.8 g, then the molecular formula of hydrocarbon is
Detailed Solution: Question 21
Detailed Solution: Question 22
The median of a grouped data is 39.8. The lower limit of the median class is 35 and the frequency is 10. The cumulative frequency of the preceding class of the median class is 34 and the total number of observations is 80. The class size of the grouped data is
Detailed Solution: Question 23
The interval in which x (> 0) must lie so that the greatest term in the expansion of (1 + x)2n has the greatest coefficient is
Detailed Solution: Question 24
Detailed Solution: Question 25
Detailed Solution: Question 26
In the given figure, OABC is a rectangle. If 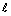 || m, what is the area of the rectangle OABC?
|| m, what is the area of the rectangle OABC?

Detailed Solution: Question 27
The number of ways in which four faces of a regular tetrahedron can be painted with four different colours is
Detailed Solution: Question 28
Detailed Solution: Question 29
Detailed Solution: Question 30


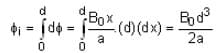
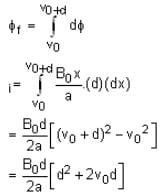
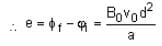
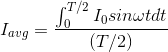
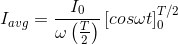
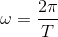 .
.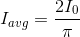



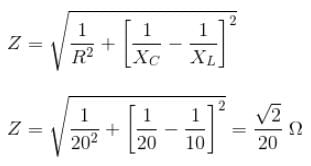
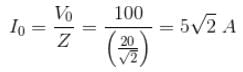
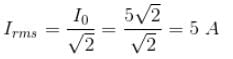
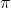 r2I, where r is the radius of the circular loop.
r2I, where r is the radius of the circular loop.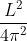
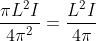
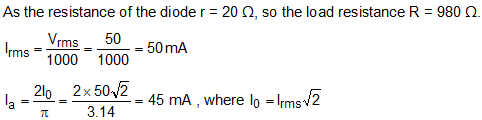
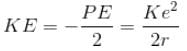


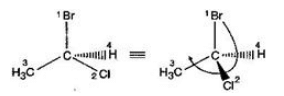
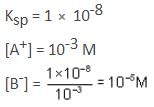
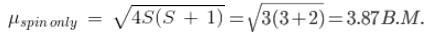
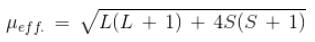
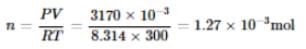
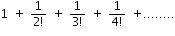 ... is
... is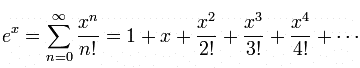
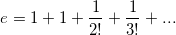
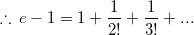
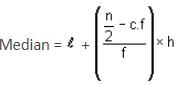
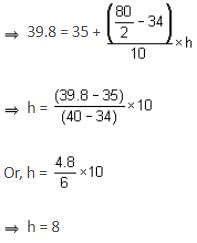
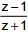 is purely imaginary, then | z | =?
is purely imaginary, then | z | =?