Chemical Kinetics - 1 - NEET Chemistry Class 12 Free MCQ Test with solutions
MCQ Practice Test & Solutions: Multiple Choice Questions (MCQs): Chemical Kinetics - 1 (18 Questions)
You can prepare effectively for NEET Chemistry Class 12 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Multiple Choice Questions (MCQs): Chemical Kinetics - 1". These 18 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 18 minutes
- - Number of Questions: 18
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following statement is true for order of a reaction?
Detailed Solution: Question 1
For the first order reaction, half life is equal to
Detailed Solution: Question 2
Straight line graph for first order reaction is obtained between
Detailed Solution: Question 3
The effect of temperature on reaction rate is given by
Detailed Solution: Question 4
The radioactive isotope used in determining the age of organic substances is
Detailed Solution: Question 5
Which among the following statement is not true for catalyst?
Detailed Solution: Question 6
A first order reaction is 50% completed in 1.26 × 1014 s. How much time would it take for 100% completion?
Detailed Solution: Question 7
Collision theory is applicable to
Detailed Solution: Question 8
Rate of reaction does not remain constant throughout because
Detailed Solution: Question 9
The unit of rate constant for a first order reaction is
Detailed Solution: Question 10
Which among the following statement is not true for rate constant of a reaction?
Detailed Solution: Question 11
Which among the following is an example of pseudo first order reaction?
Detailed Solution: Question 12
Which among the following is an example of first order reaction?
Detailed Solution: Question 13
The rate law for the reaction 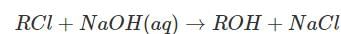 is given by rate = k[RCl]. The rate for this reaction
is given by rate = k[RCl]. The rate for this reaction
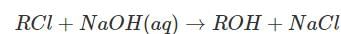 is given by rate = k[RCl]. The rate for this reaction
is given by rate = k[RCl]. The rate for this reactionDetailed Solution: Question 14
For an endothermic reaction, the minimum value for the energy of activation in terms of will be
Detailed Solution: Question 15
The temperature coefficient of most of the reactions lies between
Detailed Solution: Question 16
Following mechanism has been proposed for a reaction
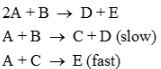
The rate law expression for the reaction is
Detailed Solution: Question 17
The expression which gives 3/4th life of first order reaction is
Detailed Solution: Question 18
54 videos|280 docs|74 tests |


