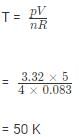States of Matter (Old NCERT) - Free MCQ Practice Test with solutions, NEET
MCQ Practice Test & Solutions: P. Bahadur Test: States of Matter (Old NCERT) (25 Questions)
You can prepare effectively for NEET Physical Chemistry for NEET with this dedicated MCQ Practice Test (available with solutions) on the important topic of "P. Bahadur Test: States of Matter (Old NCERT)". These 25 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 25 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
The average Kinetic energy and Thermal energy are proportional to the:
Detailed Solution: Question 1
Above the Critical temperature (TC) of carbon dioxide:
Detailed Solution: Question 2
A student forgot to add the reaction mixture to the round bottomed flask at 27 °C but instead he/she placed the flask on the flame. After a lapse of time, he realized his mistake, and using a pyrometer he found the temperature of the flask was 477 °C. What fraction of air would have been expelled out?
Detailed Solution: Question 3
The pressure of a 1:4 mixture of dihydrogen and dioxygen enclosed in a vessel is one atmosphere. What would be the partial pressure of dioxygen?
Detailed Solution: Question 4
The ease with which the electron cloud of a particle can be distorted is called its
Detailed Solution: Question 5
Gases have much lower density than the solids and liquids because
Detailed Solution: Question 6
The three states of matter of H2O are in equilibrium at
Detailed Solution: Question 7
Calculate the temperature of 4.0 mol of a gas occupying 5 dm3 at 3.32 bar. (R = 0.083 bar dm3K−1mol−1)
Detailed Solution: Question 8
As the temperature increases, average kinetic energy of molecules increases. What would be the effect of increase of temperature on pressure provided the volume is constant?
Detailed Solution: Question 9
Molecules in the interior of liquid experience intermolecular
Detailed Solution: Question 10
Charles’ law states that pressure remaining constant, the volume of a fixed mass of a gas is
Detailed Solution: Question 11
Surface tension decreases as the temperature is raised because
Detailed Solution: Question 12
Calculate the total number of electrons present in 1.4 g of dinitrogen gas.
Detailed Solution: Question 13
Gases possess characteristic critical temperature which depends upon the magnitude of intermolecular forces between the particles. Following are the critical temperatures of some gases. From the data what would be the order of liquefaction of these gases? Start writing the order from the gas liquefying first 
Detailed Solution: Question 14
Detailed Solution: Question 15
Gay Lussac’s law states that at constant volume, pressure of a fixed amount of a gas
Detailed Solution: Question 16
Detailed Solution: Question 17
The van der Waals Equation adjusts the measured volume
Detailed Solution: Question 18
Detailed Solution: Question 19
Capillarity results from a competition between the
Detailed Solution: Question 20
The statements for laws of chemical combinations are given below. Mark the statement which is not correct.
Detailed Solution: Question 21
Water has high surface tension and high capillarity because of
Detailed Solution: Question 22
The van der Waals Equation adjusts the measured pressure
Detailed Solution: Question 23
Atmospheric pressures recorded in different cities are as follows: Consider the above data and mark the place at which liquid will boil first. 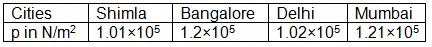
Detailed Solution: Question 24
Which curve in Figure represents the curve of ideal gas? 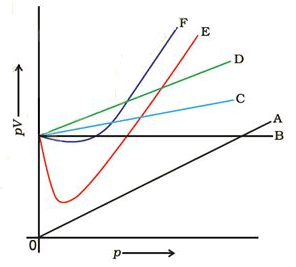
Detailed Solution: Question 25
117 videos|224 docs|237 tests |