The Solid State (Old NCERT) - Free MCQ Practice Test with solutions, JEE
MCQ Practice Test & Solutions: R.C. Mukherjee Test: The Solid State (Old NCERT) (30 Questions)
You can prepare effectively for JEE Chapter-wise Tests for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "R.C. Mukherjee Test: The Solid State (Old NCERT)". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 45 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
How many number of atoms are there in a cube based unit cell having one atom on each corner and two atoms on each body diagonal of cube -
Detailed Solution: Question 1
A metal crystallizes with a face-centered cubic lattice. The edge of the unit cell is 408 pm. The diameter of the metal atom is
Detailed Solution: Question 2
The number of atoms/molecules contained in one face centred cubic unit cell of a mono atomic substance is-
Detailed Solution: Question 3
In a face centred cubic arrangement of A & B atoms whose A atoms are at the corner of the unit cell & B atoms at the face centres. One of the A atom is missing from one corner in unit cell. The simplest formula of compound is-
Detailed Solution: Question 4
Detailed Solution: Question 5
A solid has a b.c.c. structure. If the distance of closest approach between the two atoms is 1.73 Å. The edge length of the cell is-
Detailed Solution: Question 6
In a close packed array of N spheres, the number of tetrahedral holes are-
Detailed Solution: Question 7
In a face centred cubic cell, an atom at the face centre is shared by-
Detailed Solution: Question 8
A solid XY has NaCl structure. If radius of X+ is 100 pm. What is the radius of Y- ion-
Detailed Solution: Question 9
How many atoms are there in a unit cell of Mg which forms hexagonal crystals, there being a face-centred atom in each end of the unit cell and 3 completely enclosed atoms within the unit cell-
Detailed Solution: Question 10
A solid is made of two elements X and Z. The atoms Z are in c.c.p. arrangement while atoms X occupy all the tetrahedral sites. What is the formula of the compound-
Detailed Solution: Question 11
Close packing is maximum in the crystal-
Detailed Solution: Question 12
The vacant space in b.c.c. unit cell is-
Detailed Solution: Question 13
AB crystallizes in a body centred cubic lattice with edge length 'a' equal to 387 pm. The distance between two oppositely charged ions in the lattice is
Detailed Solution: Question 14
The unit cell cube length for LiCl (just like NaCl structure) is 5.14 Å. Assuming anion-anion contact, the ionic radius for chloride ion is-
Detailed Solution: Question 15
At room temperature, sodium crystallizes in a body centred cubic lattice with a = 4.24 Å. The theoretical density of sodium (At. wt. of Na = 23) is-
Detailed Solution: Question 16
The number of octahedral void(s) per atom present in a cubic close-packed structure is
[CBSE AIPMT 2012]
Detailed Solution: Question 17
In a face centred cubic lattice the number of nearest neighbour for a given lattice point are-
Detailed Solution: Question 18
AB crystallizes in a body centred cubic lattice with edge length 'a' equal to 387 pm. The distance between two oppositively charged ions in the lattice is
Detailed Solution: Question 19
A solid AB has rock salt structure. If the edge length is 520 pm and radius of A+ is 80 pm, the radius of anion B- would be -
Detailed Solution: Question 20
A binary solid (A+ B-) has a zinc blende structure with B¯ ions constituting the lattice and A+ ions occupying 25% tetrahedral holes. The formula of solid is-
Detailed Solution: Question 21
A certain metal crystallises in a simple cubic structure. At a certain temperature, it arranges to give a body centred structure. In this transition, the density of the metal-
Detailed Solution: Question 22
For the structure given below the site marked as S is a-
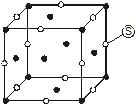
Detailed Solution: Question 23
Detailed Solution: Question 24
Detailed Solution: Question 25
A solid having no definite shape is called-
Detailed Solution: Question 26
The mass of a unit cell of CsCl corresponds to-
Detailed Solution: Question 27
Close packing is maximum in the crystal lattice of-
Detailed Solution: Question 28
The structure of MgO is similar to NaCl. The coordination number of Mg is-
Detailed Solution: Question 29
Each unit cell of NaCl consists of 14 chlorine atoms and-
Detailed Solution: Question 30
446 docs|929 tests |


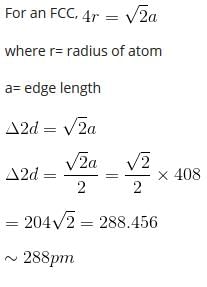

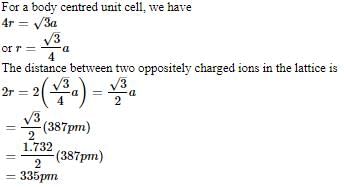 option B
option B 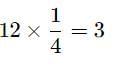
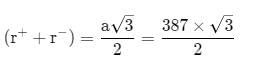 = 335.15 pm
= 335.15 pm