Thermodynamics - Free MCQ Practice Test with solutions, NEET Chemistry
MCQ Practice Test & Solutions: R.C. Mukherjee Test: Thermodynamics (25 Questions)
You can prepare effectively for NEET Chemistry Class 11 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "R.C. Mukherjee Test: Thermodynamics". These 25 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 35 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
If Vf is the final volume and Vi is the initial volume and pex the external pressure the work done can be calculated by
Detailed Solution: Question 1
Detailed Solution: Question 2
Detailed Solution: Question 3
Spontaneity in the context of chemical thermodynamics means
Detailed Solution: Question 4
Enthalpy of combustion of carbon to CO2is –393.5 kJ mol−1. Calculate the heat released upon formation of 35.2 g of CO2from carbon and dioxygen gas.
Detailed Solution: Question 5
The volume of gas is reduced to half from its original volume. The specific heat will
Detailed Solution: Question 6
A cylinder confines 2.00 L gas under a pressure of 1.00 atm. The external pressure is also 1.00 atm. The gas is heated slowly, with the piston sliding freely outward to maintain the pressure of the gas close to 1.00 atm. Suppose the heating continues until a final volume of 3.50 L is reached. Calculate the work done on the gas.
Detailed Solution: Question 7
A spherical constant temperature heat source of radius r1 is at the center of a uniform solid sphere of radius r2. The rate at which heat is transferred through the surface of the sphere is proportional to
Detailed Solution: Question 8
For a process from state 1 to state 2, heat transfer in a reversible process is given by
Detailed Solution: Question 9
Suppose a 10.00-kg mass drops through a height difference of 3.00 m, and the resulting work is used to turn a paddle in 200.0 g water, initially at 15.00∘C. The final water temperature is found to be 15.35∘C. Assuming that the work done is used entirely to increase the water temperature, calculate the conversion factor between joules and calories.
Detailed Solution: Question 10
The molar heat capacity of a substance is the
Detailed Solution: Question 11
If change in Gibbs energy ΔG is negative (< 0)
Detailed Solution: Question 12
Calculate the standard enthalpy of formation of CH3OH(l) from the following data:
CH3OH(I)+3/2O2(g)→CO2(g)+2H2O(l); △rHθ=−726kJmol−1
C(graphite)+O2(g)→CO2(g); △cHθ=−393kJmol−1
H2(g)+1/2O2(g)→H2O(l); △fHθ=−286kJmol−1
Detailed Solution: Question 13
During complete combustion of one mole of butane, 2658 kJ of heat is released. The thermochemical reaction for above change is ΔfU0 of formation of CH4 (g) at certain temperature is –393 kJ mol−1. The value of ΔfH0is
Detailed Solution: Question 14
In an adiabatic process, no transfer of heat takes place between system and surroundings. Choose the correct option for free expansion of an ideal gas under adiabatic condition from the following.
Detailed Solution: Question 15
The relationship between Cp and CV for an ideal gas is
Detailed Solution: Question 16
Detailed Solution: Question 17
A thermodynamic state function is a physical quantity
Detailed Solution: Question 18
Detailed Solution: Question 19
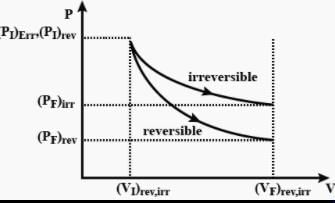
The pressure-volume work for an ideal gas can be calculated by using the expression 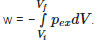 The work can also be calculated from the pV– plot by using the area under the curve within the specified limits. When an ideal gas is compressed (a) reversibly or (b) irreversibly from volume Vi to Vf . choose the correct option.
The work can also be calculated from the pV– plot by using the area under the curve within the specified limits. When an ideal gas is compressed (a) reversibly or (b) irreversibly from volume Vi to Vf . choose the correct option.
Detailed Solution: Question 20
Suppose that 1.00 kJ of heat is transferred to 2.00 mol argon (at 298 K, 1 atm). What will the final temperature Tf be if the heat is transferred at constant volume?
Detailed Solution: Question 21
Detailed Solution: Question 22
For the process to occur under adiabatic conditions, the correct condition is:
Detailed Solution: Question 23
For an isolated system, ΔU = 0, what will be ΔS?
Detailed Solution: Question 24
The entropy change can be calculated by using the expression 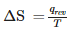 When water freezes in a glass beaker, choose the correct statement amongst the following :
When water freezes in a glass beaker, choose the correct statement amongst the following :
Detailed Solution: Question 25
98 videos|243 docs|71 tests |