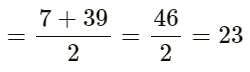Science Olympiad Test: Periodic Classification of Elements- 2 - Free MCQ
MCQ Practice Test & Solutions: Science Olympiad Test: Periodic Classification of Elements- 2 (15 Questions)
You can prepare effectively for Class 10 Olympiad Preparation for Class 10 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Science Olympiad Test: Periodic Classification of Elements- 2". These 15 questions have been designed by the experts with the latest curriculum of Class 10 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 15 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
The element with atomic number 14 is hard and forms acidic oxide and a covalent halide. To which of the following categories does the element belong?
Detailed Solution: Question 1
What type of oxide would Eka-aluminium form?
Detailed Solution: Question 2
Which of the following does not increase while moving down the group of the periodic table?
Detailed Solution: Question 3
Which of the following elements will form an acidic oxide?
Detailed Solution: Question 4
Arrange the following elements in the order of the their increasing non-metallic character Li, O, C, Be, F
Detailed Solution: Question 5
Elements A, B and C Constitute a Dobereiner’s triad. If the atomic mass of element A is 7 and that of element C is 39, then what is the atomic mass of element B?
Detailed Solution: Question 6
X and Y are two elements having similar properties which obey Newland’s Law of Octaves. The minimum and maximum number of elements in between X and Y, respectively are
Detailed Solution: Question 7
The two elements for which Mendeleev left blank places in his original periodic table were
Detailed Solution: Question 8
Which of the following statement(s) about the modern periodic table are incorrect
(i) The elements in the modern periodic table are arranged on the basis of their decreasing atomic number.
(ii) Isotopes are placed in adjoining group(s) in the periodic table
(iii) The elements in the modern period table are arranged on the basis of their increasing atomic masses.
(iv) The elements in the modern periodic table are arranged on the basis of their increasing atomic number.
(i) The elements in the modern periodic table are arranged on the basis of their decreasing atomic number.
(ii) Isotopes are placed in adjoining group(s) in the periodic table
(iii) The elements in the modern period table are arranged on the basis of their increasing atomic masses.
(iv) The elements in the modern periodic table are arranged on the basis of their increasing atomic number.
Detailed Solution: Question 9
The elements A, B, C, D and E have atomic number 9, 11, 17, 12 and 13 respectively. Which pair of elements belong to the same group?
Detailed Solution: Question 10
Which of the following statements about the modern period table is correct?
Detailed Solution: Question 11
Which one of the following elements exhibit maximum number of valence electrons?
Detailed Solution: Question 12
Which of the following gives the correct increasing order of the atomic radii of O, F and N?
Detailed Solution: Question 13
Which of the following are the characteristics of isotopes of an element?
(i) Isotopes of an element have same atomic masses
(ii) Isotopes of an element have same atomic number
(iii) Isotopes of an element show same chemical properties
(iv) Isotopes of an element show same physical properties
(i) Isotopes of an element have same atomic masses
(ii) Isotopes of an element have same atomic number
(iii) Isotopes of an element show same chemical properties
(iv) Isotopes of an element show same physical properties
Detailed Solution: Question 14
Three elements B, Si and Ge are
Detailed Solution: Question 15
70 videos|236 docs|187 tests |