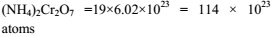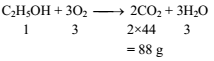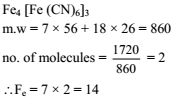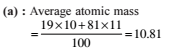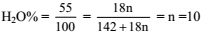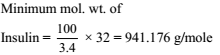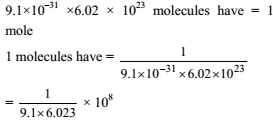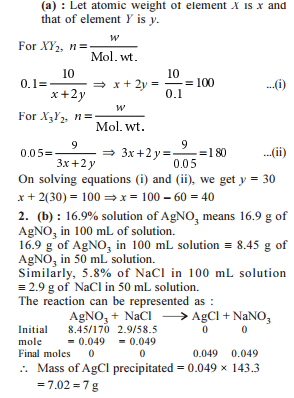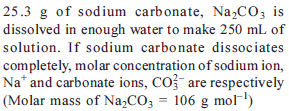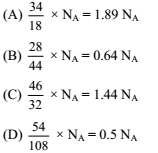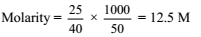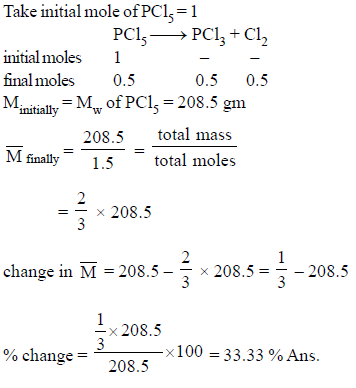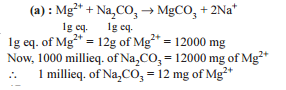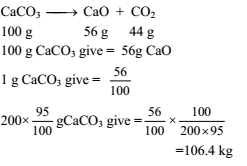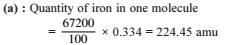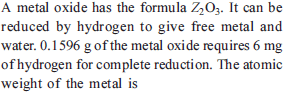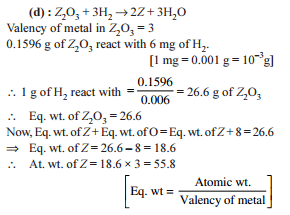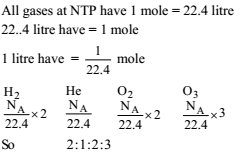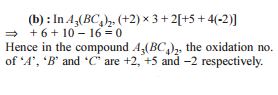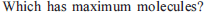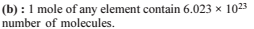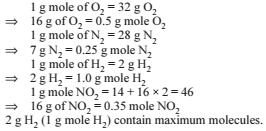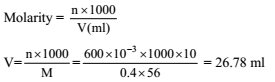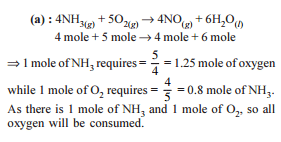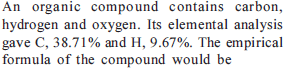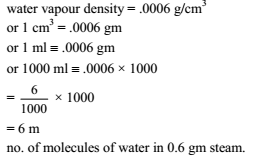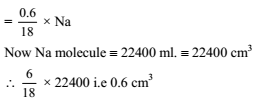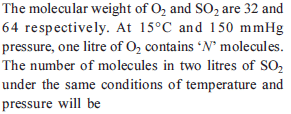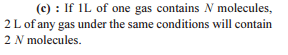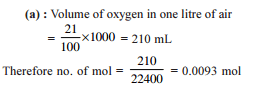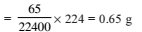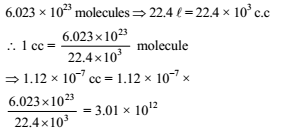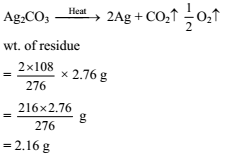Some Basic Concepts of Chemistry - 1 - Free MCQ Test with solutions
MCQ Practice Test & Solutions: Some Basic Concepts of Chemistry - 1 (30 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Some Basic Concepts of Chemistry - 1". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Total number of atoms of all elements present in 1 mole of ammonium dichromate is ?
Detailed Solution: Question 1
If one mole of ethanol (C2H5OH) completely burns to carbon dioxide and water, the weight of carbon dioxide formed is about -
(C2H5OH + 3O2 → 2CO2 + 3H2O)
(C2H5OH + 3O2 → 2CO2 + 3H2O)
Detailed Solution: Question 2
The iron atoms in 1720 amu of ferric ferrocyanide is .....................
Detailed Solution: Question 3
Detailed Solution: Question 4
The hydrated salt Na2SO4.nH2O, undergoes 55% loss in weight on heating and becomes anhydrous. The value of n will be
Detailed Solution: Question 5
Insulin contains 3.4% sulphur. What will be the minimum molecular weight of insulin ?
Detailed Solution: Question 6
How many moles of electron weight one kilogram ?
Detailed Solution: Question 7
Detailed Solution: Question 8
Detailed Solution: Question 9
The largest number of molecules is present in
Detailed Solution: Question 10
25 g of NaOH is dissolved in 50 mL of water. The molarity of the solution is
Detailed Solution: Question 11
Calculate percentage change in Mavg of the mixture, if PCl5 undergo 50% decomposition.
PCl5 → PCl3 + Cl2
Detailed Solution: Question 12
Detailed Solution: Question 13
Calculate the weight of lime (CaO) obtained by heating 200 kg of 95% pure lime stone (CaCO3).
Detailed Solution: Question 14
Detailed Solution: Question 15
Detailed Solution: Question 16
A carbon compound containing carbon and oxygen has molar mass equal to 288. On analysis it is found to contain 50% by mass of
each element. Therefore molecular formula of the compound is -
each element. Therefore molecular formula of the compound is -
Detailed Solution: Question 17
Four one litre flasks are separately filled with the gases hydrogen, helium, oxygen and ozone at the same room temperature and pressure.
The ratio of total number of atoms of these gases present in the different flasks would be
The ratio of total number of atoms of these gases present in the different flasks would be
Detailed Solution: Question 18
Detailed Solution: Question 19
Detailed Solution: Question 20
Detailed Solution: Question 21
What volume of 0.4-M FeCl3.6H2O will contain 600 mg of Fe3+ ?
Detailed Solution: Question 22
Detailed Solution: Question 23
Detailed Solution: Question 24
At 100ºC and 1 atm, if the density of liquid water is 1.0 g cm–3 and that of water vapor is 0.0006 g cm–3, then the volume occupied by water molecules in 1 litre of steam at that temperature is -
Detailed Solution: Question 25
Detailed Solution: Question 26
Detailed Solution: Question 27
Detailed Solution: Question 28
Volume of a gas at NTP is 1.12 × 10–7 c.c. The number of molecules in it will be -
Detailed Solution: Question 29
Detailed Solution: Question 30
332 videos|699 docs|300 tests |