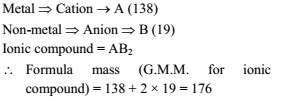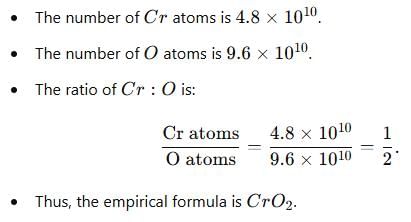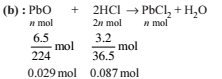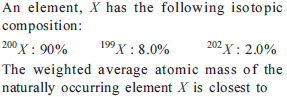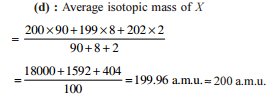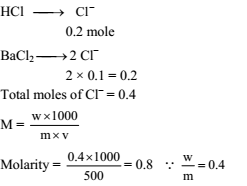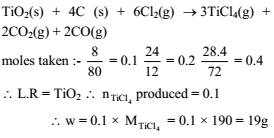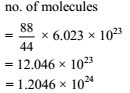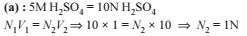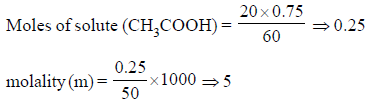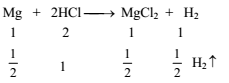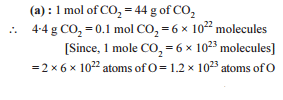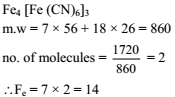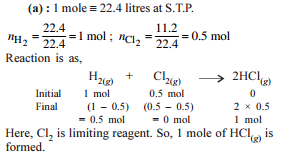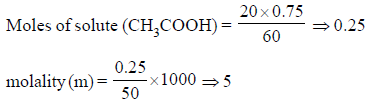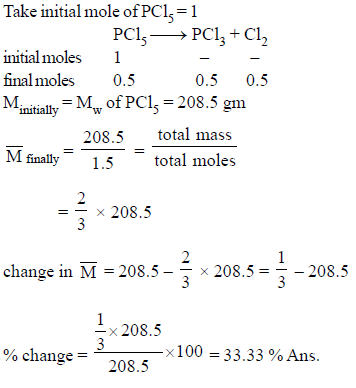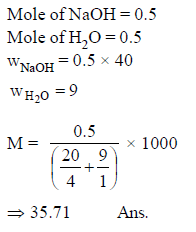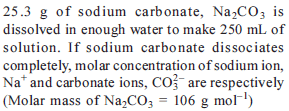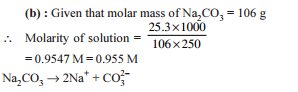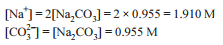Some Basic Concepts of Chemistry - 2 - Free MCQ Test with solutions
MCQ Practice Test & Solutions: Some Basic Concepts of Chemistry - 2 (30 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Some Basic Concepts of Chemistry - 2". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
The volume of 1.204 × 1024 molecules of water at 4ºC is …………… (ml)
Detailed Solution: Question 2
There are two common oxides of Sulphur, one of which contains 50% O2 by weight, the other almost exactly 60%. The weights of sulphur
which combine with 1 g of O2 (fixed) are in the ratio of -
which combine with 1 g of O2 (fixed) are in the ratio of -
Detailed Solution: Question 3
Detailed Solution: Question 4
Detailed Solution: Question 5
Detailed Solution: Question 6
In an ionic compound moles ratio of cation to anion is 1 : 2. If atomic masses of metal and non-metal respectively are 138 and 19, then
correct statement is -
correct statement is -
Detailed Solution: Question 7
The number of atoms of Cr and O are 4.8 × 1010 and 9.6 × 1010 respectively. Its empirical formula is -
Detailed Solution: Question 8
Iron forms two oxides, in first oxide 56 gram. Iron is found to be combined with 16 gram oxygen and in second oxide 112 gram iron is
found to be combined with 48 gram oxygen. This data satisfy the law of -
found to be combined with 48 gram oxygen. This data satisfy the law of -
Detailed Solution: Question 9
 How many moles of lead (II) chloride will be formed from the reaction between 6.5 g of PbO and 3.2 g of HCl?
How many moles of lead (II) chloride will be formed from the reaction between 6.5 g of PbO and 3.2 g of HCl?
Detailed Solution: Question 10
Detailed Solution: Question 11
0.2 mole of HCl and 0.1 mole of barium chloride were dissolved in water to produce a 500-mL solution. The molarity of the Cl– ions is -
Detailed Solution: Question 12
A vessel contains 8 gram TiO2, 2.4 gram carbon and 28.4 gram Cl2. Maximum mass of TiCl4 which can be produced is -
3TiO2(s) + 4C(s) + 6Cl2(g) → 3TiCl4(g) + 2CO2(g) + 2CO(g)
(Consider reaction goes to completion ; atomic mass of Ti = 48)
3TiO2(s) + 4C(s) + 6Cl2(g) → 3TiCl4(g) + 2CO2(g) + 2CO(g)
(Consider reaction goes to completion ; atomic mass of Ti = 48)
Detailed Solution: Question 13
The mole fraction of a given sample of I2 in C6H6 is 0.2. The molality of I2 in C6H6 is -
Detailed Solution: Question 14
The number of molecules present in 88 g of CO2 (Relative molecular mass of CO2 = 44)
Detailed Solution: Question 15
Detailed Solution: Question 16
When 20 ml of pure acetic acid (density = 0.75 gm ml–1) is mixed with 50 gm of water (density = 1gm ml–1) at a certain temperature. Calculate the molality of acetic acid in the final solution.
Detailed Solution: Question 17
If NA is Avogadro number, then the number of valence electrons in 4.2 g of N3– ions is -
Detailed Solution: Question 18
12g of Mg (atm. mass 24) will react completely with acid to give -
Detailed Solution: Question 19
Detailed Solution: Question 20
The iron atoms in 1720 amu of ferric ferrocyanide is .....................
Detailed Solution: Question 21
Detailed Solution: Question 22
Detailed Solution: Question 23
When 20 ml of pure acetic acid (density = 0.75 gm ml–1) is mixed with 50 gm of water (density = 1gm ml–1) at a certain temperature. Calculate the molality of acetic acid in the final solution.
Detailed Solution: Question 24
Calculate percentage change in Mavg of the mixture, if PCl5 undergo 50% decomposition.
PCl5 → PCl3 + Cl2
Detailed Solution: Question 25
Molarity of aqueous NaOH solution will be, if mole fraction of NaOH in the solution is 0.5. [Given : density of pure NaOH = 4 gm/ml]
Detailed Solution: Question 26
Detailed Solution: Question 27
2 moles of Al react with 3 moles of Cl2 to form AlCl3. What will be the limiting reagent, and how many moles of AlCl3 will be formed?
Detailed Solution: Question 28
A compound contains 50% sulfur and 50% oxygen by mass. What is the empirical formula of the compound?
Detailed Solution: Question 29
A sample contains 3 mol of N2 and 2mol of H2. What is the total number of molecules in the sample?
Detailed Solution: Question 30
332 videos|699 docs|300 tests |