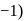Balancing of redox reactions, Types reactions - Free MCQ Practice Test
MCQ Practice Test & Solutions: Test: Balancing of redox reactions, Types of redox reactions (10 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Balancing of redox reactions, Types of redox reactions". These 10 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following represents a redox reaction?
Detailed Solution: Question 1
In the disproportionation reaction 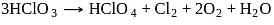 , the equivalent mass of the oxidizing agent is (molar mass of
, the equivalent mass of the oxidizing agent is (molar mass of 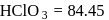 )
)
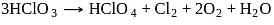 , the equivalent mass of the oxidizing agent is (molar mass of
, the equivalent mass of the oxidizing agent is (molar mass of 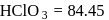 )
)Detailed Solution: Question 2
Which of the following is only a redox reaction but not a disproportionation reaction?
Detailed Solution: Question 3
Which of the following do not show disproportionation reaction? 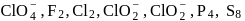 , and
, and 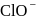
Detailed Solution: Question 4
Determine the oxidation number of the underlined atom in 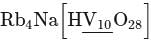
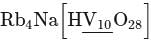
Detailed Solution: Question 5
The oxidation state of 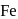 in
in 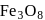 is
is
Detailed Solution: Question 6
Oxidation state of sulphur in anions 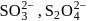 and
and 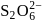 increases in the orders:
increases in the orders:
Detailed Solution: Question 7
Amongst the following, identify the species with an atom in 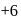 oxidation state:
oxidation state:
Detailed Solution: Question 8
Point out the correct statement of the following about 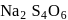
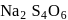
Detailed Solution: Question 9
A mixture of potassium chlorate, oxalic acid and sulphuric acid is heated. During the reaction which element undergoes maximum change in the oxidation number?
Detailed Solution: Question 10
335 videos|697 docs|300 tests |


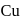 is
is 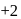 in both reactant
in both reactant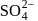 ion does not change. In option (d) is a redox reaction.
ion does not change. In option (d) is a redox reaction.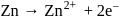 (Oxidation)
(Oxidation)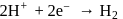 (Reduction)
(Reduction)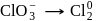
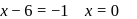
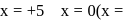 oxidation number
oxidation number 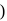
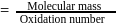


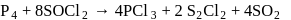 involves change of oxidation state of
involves change of oxidation state of 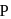 from 0 to +3 and that of
from 0 to +3 and that of 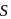 from +4 to +2 .
from +4 to +2 .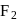 being most electronegative element cannot exhibit any positive oxidation state. In
being most electronegative element cannot exhibit any positive oxidation state. In 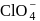 chlorine is present in its highest oxidation state i.e
chlorine is present in its highest oxidation state i.e 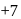 . Therefore it does not show disproportionation reaction.
. Therefore it does not show disproportionation reaction.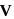 be
be 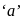 , then
, then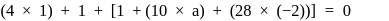
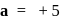
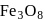
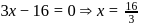
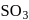
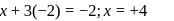
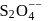
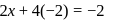
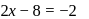
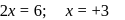
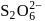

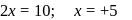
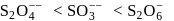
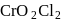
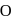 . No. of
. No. of 
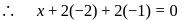
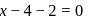
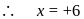
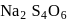 has the structure :
has the structure :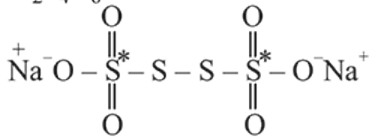
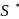 atoms are
atoms are 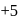 each and that of other two S atoms is zero each.
each and that of other two S atoms is zero each.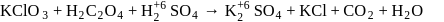
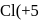 to
to