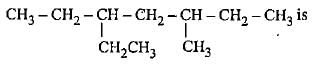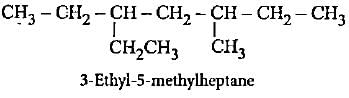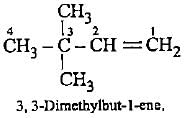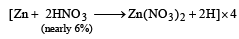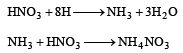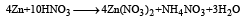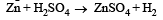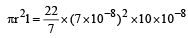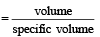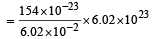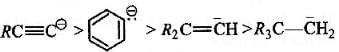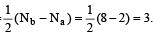Test : Chemistry Class 11 Mini Mock - 5 NEET MCQs & solutions - Free
Full Mock Test & Solutions: Test : Chemistry Class 11 Mini Mock - 5 (45 Questions)
You can boost your NEET 2026 exam preparation with this Test : Chemistry Class 11 Mini Mock - 5 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of NEET 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 45 minutes
- - Total Questions: 45
- - Analysis: Detailed Solutions & Performance Insights
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
Which orbital gives an electron, a greater probability being found close to the nucleus?
Detailed Solution: Question 1
C2H4 + xO2 -> 2CO2 + yH2O, what is the value of x + y?
Detailed Solution: Question 2
The empirical formula of a compound is CH2O2.What could be its molecular formula?
Detailed Solution: Question 3
Detailed Solution: Question 4
Spin quantum number with two spin states of the electron represented by two arrows, ↑ (spin up) and ↓ (spin down) was introduced to account for
Detailed Solution: Question 5
When H+ attacks CH3 – CH = CH2 , carbonation which is more stable is
Detailed Solution: Question 6
Formula mass is used for which of the following substances?
Detailed Solution: Question 7
In which of the following species the underlined carbon is having sp3 − hybridization
Detailed Solution: Question 8
Detailed Solution: Question 9
In oxygen difluoride (OF2) and dioxygen difluoride(O2F2), the oxygen is assigned an oxidation number of
Detailed Solution: Question 10
Zn gives H2 gas with H2SO4 and HCl but not with HNO3 because [2002]
Detailed Solution: Question 11
The major product of the following reaction is
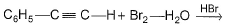
Fill in the blanks by picking the correct option.
There are _____ groups and _____ periods in the extended form of periodic table. The group, all members of which are in gaseous state under ordinary conditions is _____ group. Most electropositive elements belong to _____ group.
There are _____ groups and _____ periods in the extended form of periodic table. The group, all members of which are in gaseous state under ordinary conditions is _____ group. Most electropositive elements belong to _____ group.
Detailed Solution: Question 13
Molecule MX3 (atomic number M < 21) has zero dipole moment, the sigma bonding orbitals used by M are
Detailed Solution: Question 14
Pick out the pair of species having identical shapes for both the molecules.
Detailed Solution: Question 15
Specific volume of cylindrical virus particle is 6.02 × 10–2 cc/gm. whose radius and length 7 Å & 10 Å respectively. If NA = 6.02 × 1023, find molecular weight of virus [2001]
Detailed Solution: Question 16
Which of the following statements is false?
Detailed Solution: Question 17
Direction (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.
Q. The reversible expansion of an ideal gas under adiabatic and isothermal conditions is shown in the following figure. Select correct statement.
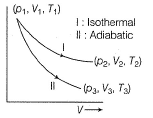
[IIT JEE 2012]
Detailed Solution: Question 18
The stability of carbanions in the following :


is in the order of : [2008]


Detailed Solution: Question 19
Find the correct order for relative energies of the ethane conformations
Detailed Solution: Question 20
The two effective drugs which act as life-saving drugs for cancer therapy and AIDS victims respectively are:
Detailed Solution: Question 21
Detailed Solution: Question 22
The ground state electronic configuration of valence shell electrons in nitrogen molecule (N2) is written as KK 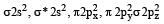 Bond order in nitrogen molecule is [1995]
Bond order in nitrogen molecule is [1995]
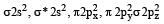 Bond order in nitrogen molecule is [1995]
Bond order in nitrogen molecule is [1995]Detailed Solution: Question 23
Direction (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
The molecular formula C5H12 contains how many isomeric alkanes?
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Detailed Solution: Question 24
The distinction between atoms and molecules was made by:
Detailed Solution: Question 25
Detailed Solution: Question 26
A sample containing 1.0 mole of an ideal gas is expanded isothermally and reversibly to ten time of its original volume, in two separate experiments. The expansion is carried out 300 K and at 600 K, respectively. Choose the correct option.
Detailed Solution: Question 27
The atom with the given atomic number Z=17, and the atomic mass A=35.5 is
Detailed Solution: Question 28
How many different stereoisomers exist for 1-chloro-2-(3-chlorocyclobutyl) ethene?
Detailed Solution: Question 29
Which of the following compounds is more easily oxidised to a carbonyl when treated with MnO2?
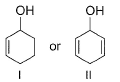
Detailed Solution: Question 30
98 videos|243 docs|71 tests |