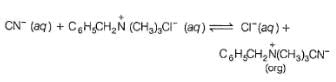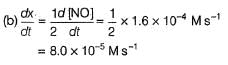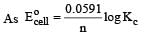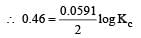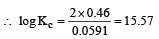Test : Chemistry Class 12 Mini Mock - 5 NEET MCQs & solutions - Free
Full Mock Test & Solutions: Test : Chemistry Class 12 Mini Mock - 5 (45 Questions)
You can boost your NEET 2026 exam preparation with this Test : Chemistry Class 12 Mini Mock - 5 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of NEET 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 45 minutes
- - Total Questions: 45
- - Analysis: Detailed Solutions & Performance Insights
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
Detailed Solution: Question 1
The number of moles of oxygen in one litre of air containing 21% oxygen by volume, in standard conditions, is [1995]
Detailed Solution: Question 2
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.
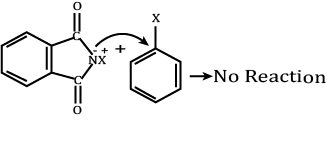
Assertion (A): Aromatic 1° amines can be prepared by Gabriel Phthalimide synthesis.
Reason (R): Aryl halides do not undergo nucleophilic substitution with anion formed by phthalimide.
Detailed Solution: Question 3
Detailed Solution: Question 4
Detailed Solution: Question 5
Detailed Solution: Question 6
The reduction potential of an element A is -2.71V. What can be concluded from this?
Detailed Solution: Question 7
A foreign substance that increase the speed of a chemical reaction is called
Detailed Solution: Question 8
Which of the following statement is not correct? [2001]
Detailed Solution: Question 9
Which among the following is an example of photochemistry used in our daily life?
Detailed Solution: Question 10
Which base is present in RNA but not in DNA?
Detailed Solution: Question 11
Only One Option Correct Type
This section contains multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q.
Cl2 gas is passed into a solution containing KF, Kl and KBr, and CHCI3 is added. There is a colour in CHCI3 (lower) layer. It is due to
Detailed Solution: Question 12
Which of the following is a tertiary halogenoalkanes?
Detailed Solution: Question 13
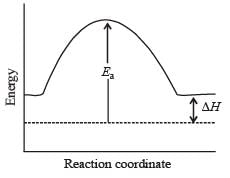
For an endothermic reaction, energy of activation is Ea and enthalpy of reaction of ΔH (both of these in kJ/mol). Minimum value of Ea will be. [2010]
Detailed Solution: Question 14
What is the correct increasing order of reactivity of the following in the SN2 reaction?

Detailed Solution: Question 15
The CFSE for octahedral [CoCI6]4- is 18000 cm-1. Then the CFSE for tetrahedral [CoCI4]2- will be
Detailed Solution: Question 16
Arrange the following alcohols, hydrocarbon and ether in order of their increasing boiling points Pentan – 1 – ol, n – butane, pentanal, ethoxyethane.
Detailed Solution: Question 17
The element used in the production of photocells and solar cells and also used in xerox machines is
Detailed Solution: Question 18
The best method for the separation of naphthalene and benzoic acid from their mixture is
Detailed Solution: Question 19
Ketones are reduced to the corresponding alcohols by catalytic hydrogenation to form
Detailed Solution: Question 20
Which of the following fertilizers has the highest nitrogen percentage ? [1993]
Detailed Solution: Question 21
General electronic configuration of lanthanides is[2002]
Detailed Solution: Question 22
The relative lowering of the vapour pressure is equal to the ratio between the number of [1991]
Detailed Solution: Question 23
(i) CH3 – CH2 – Br + NaOH  CH3 – CH2 – OH + NaBr → reaction ... (i)
CH3 – CH2 – OH + NaBr → reaction ... (i)
(ii) 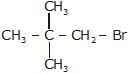 + NaOH
+ NaOH 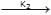 (CH3)3 C – CH2 – OH + NaBr → reaction ... (ii)
(CH3)3 C – CH2 – OH + NaBr → reaction ... (ii)
K1 & K2 are rate constant for above reaction correct relation is
Detailed Solution: Question 24
In the following two phase reaction, catalyst works by
C6H5CH2Br + KCN 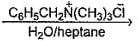 C6H5CH2CN + KBr
C6H5CH2CN + KBr
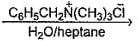 C6H5CH2CN + KBr
C6H5CH2CN + KBrDetailed Solution: Question 25
The rate of formation of NO(g) in the reaction, 2NOBr(g) → 2NO(g) + Br2(g) was reported as 1.6 x 10-4 Ms-1. Thus, rate of the reaction is
Detailed Solution: Question 26
Which one of the following arrangements does not give the correct picture of the trends indicated against it ? [2008]
Detailed Solution: Question 27
Harmful UV radiations emitted from the sun are prevented from reaching the earth by the presence of ozone in the
Detailed Solution: Question 28
The equilibrium constant of the reaction:
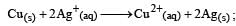
E° = 0.46 V at 298 K is [2007]
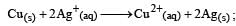
Detailed Solution: Question 29
Which of the following is a negative ligand?
Detailed Solution: Question 30
54 videos|290 docs|74 tests |


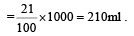
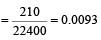

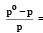 mole fraction of solute
mole fraction of solute