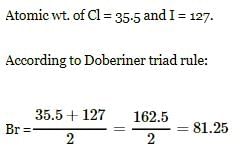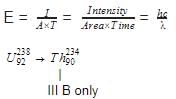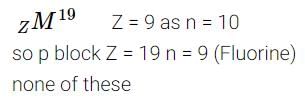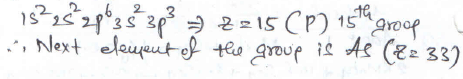Classification of Elements & Periodicity in Properties - 2 - Free MCQ Practice
MCQ Practice Test & Solutions: Test: Classification of Elements & Periodicity in Properties - 2 (30 Questions)
You can prepare effectively for NEET NCERT Based Tests for NEET with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Classification of Elements & Periodicity in Properties - 2". These 30 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following is not a Dobereiner triad -
Detailed Solution: Question 1
Which of the following set of elements obeyes Newland’s octave rule -
Detailed Solution: Question 2
Which of the following will not form crystalline structure with oppositely charged ions
Detailed Solution: Question 3
Which is not anomalous pair of elements in the Medeleeves periodic table -
Detailed Solution: Question 4
Elements in the same vertical group of the periodic table have same
Detailed Solution: Question 5
The places that were left empty by Mendeleev were, for -
Detailed Solution: Question 6
Which of the following pairs of elements do not follow octave rule -
Detailed Solution: Question 7
Elements which occupied position in the lother meyer curve, on the peaks, were -
Detailed Solution: Question 8
Modern periodic table is based on atomic no. experiments which proved importance of at no. was -
Detailed Solution: Question 9
Atomic no. is the base of -
(i) Lother meyer curve
(ii) Newland octave rule
(iii) Modern periodic table
(iv) Doeberiener triad rule
(v) Long form of periodic table
(i) Lother meyer curve
(ii) Newland octave rule
(iii) Modern periodic table
(iv) Doeberiener triad rule
(v) Long form of periodic table
Detailed Solution: Question 10
Atomic wt. or Cl = 35.5 and of I = 127. According to doeberiner triad rule, At. wt. of Br will be -
Detailed Solution: Question 11
The elements of groups, 1, 2, 13, 14, 15, 16 and 17 are collectively called -
Detailed Solution: Question 12
Justification of putting H in VII A group is -
Detailed Solution: Question 13
The discovery of which of the following group of elements gave a death blow to the Newlands Law -
Detailed Solution: Question 14
Which of the following pair of elements follows Newland’s octave rule -
Detailed Solution: Question 15
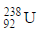 (IIIB) changes to
(IIIB) changes to 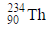 by emission of α-particle. Daughter element will be in -
by emission of α-particle. Daughter element will be in -
Detailed Solution: Question 16
From the list given below, elements which belongs to the same group or sub-group are -
Detailed Solution: Question 17
The name ‘Rare earths’ is used for -
Detailed Solution: Question 18
Z/e ratio for N3-, O2- and F- respectively will be -
Detailed Solution: Question 19
There are 10 neutrons in the nucleus of the element zM19. It belongs to -
Detailed Solution: Question 20
Which of the following is not an actinoid?
Detailed Solution: Question 21
the electronic configuration of an element is 1s2 2s2 2p6 3s2 3p3. the atomic number of the element which is just below the above element in the periodic table is ________
Detailed Solution: Question 22
The number of elements in 5th and 6th period of periodic table are respectively -
Detailed Solution: Question 23
Atomic number of Ag is 47. In the same group the atomic number of elements placed above and below Ag will be -
Detailed Solution: Question 24
Atomic number 15, 33, 51 represents the following family -
Detailed Solution: Question 25
The atom having the valence shell electronic configuration 4s2 4p2 would be in -
Detailed Solution: Question 26
The number of elements know at that time when Mendeleev arranged them in the periodic table was-
Detailed Solution: Question 27
As applied to periodic table, which of the following sets include only magic numbers -
Detailed Solution: Question 28
In the general electronic configuration -
(n - 2)f1-14 (n - 1)d0-1 ns2, if value of n = 7 the configuration will be -
Detailed Solution: Question 29
The correct order of atomic size of C, N, P, S follows the order -
Detailed Solution: Question 30