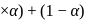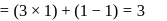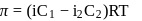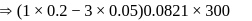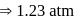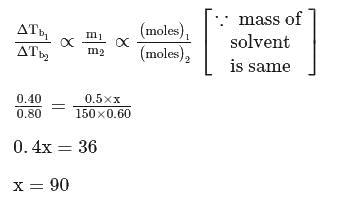Colligative Properties and Abnormal Molecular Masses - Free MCQ Practice
MCQ Practice Test & Solutions: Test: Colligative Properties and Abnormal Molecular Masses (15 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Colligative Properties and Abnormal Molecular Masses". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
What is the van't Hoff factor of Ferric Sulphate (Assume 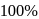 ionization)
ionization)
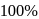 ionization)
ionization)Detailed Solution: Question 1
Relative lowering of vapour pressure is directly proportional to
Detailed Solution: Question 2
When common salt is dissolved in water:
Detailed Solution: Question 3
Camphor is used as solvent to determine the molecular mass of non-volatile solute by Rast method because for camphor
Detailed Solution: Question 4
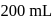 of an aqueous solution of a protein contains its
of an aqueous solution of a protein contains its 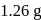 . The osmotic pressure of this solution at
. The osmotic pressure of this solution at 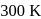 is found to be
is found to be 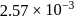 bar. The molar mass of protein will be
bar. The molar mass of protein will be 
Detailed Solution: Question 5
The difference between the boiling point and freezing point of an aqueous solution containing sucrose (molecular 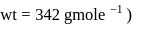 in
in 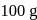 of water is
of water is 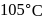 . If
. If 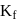 and
and 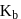 of water are
of water are 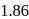 and
and 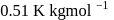 respectively, the weight of sucrose in the solution is about
respectively, the weight of sucrose in the solution is about
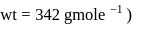 in
in 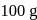 of water is
of water is 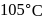 . If
. If 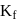 and
and 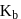 of water are
of water are 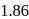 and
and 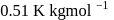 respectively, the weight of sucrose in the solution is about
respectively, the weight of sucrose in the solution is aboutDetailed Solution: Question 6
A solution containing 1.8 g of a compound (empirical formula CH2O) in 40 g of water is observed to freeze at −0.465∘C. The molecular formula of the compound is (Kf of water = 1.86 kg K mol−1)
Detailed Solution: Question 7
Freezing point of an aqueous solution is 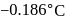 . If the values of
. If the values of 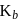 and
and 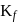 of water are respectively
of water are respectively 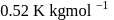 and
and 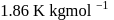 , then the elevation of boiling point of the solution in
, then the elevation of boiling point of the solution in 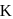 is
is
Detailed Solution: Question 8
If the elevation in boiling point of a solution of 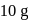 of solute
of solute 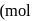 . wt.
. wt. 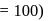 in
in 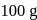 of water is
of water is 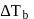 , the ebullioscopic constant of water is
, the ebullioscopic constant of water is
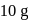 of solute
of solute 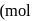 . wt.
. wt. 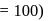 in
in 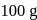 of water is
of water is 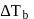 , the ebullioscopic constant of water is
, the ebullioscopic constant of water isDetailed Solution: Question 9
Which one of the following statements is incorrect?
Detailed Solution: Question 10
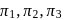 and
and 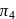 atm are the osmotic pressures of
atm are the osmotic pressures of 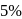 (mass/volume) solutions of urea, fructose, sucrose and
(mass/volume) solutions of urea, fructose, sucrose and 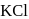 respectively at certain temperature. The correct order of their magnitudes is :
respectively at certain temperature. The correct order of their magnitudes is :
Detailed Solution: Question 11
Which of the following has been arranged in the increasing order of freezing point?
Detailed Solution: Question 12
1.0 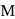 aqueous solution of
aqueous solution of 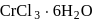 freezes at
freezes at 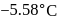 . Assuming complete ionization of the hydrated complex, which of the following isomers conforms to the observation
. Assuming complete ionization of the hydrated complex, which of the following isomers conforms to the observation 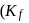 for water
for water 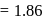
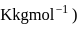 ?
?
Detailed Solution: Question 13
A living cell contains a solution which is isotonic with 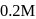 glucose solution. What osmotic pressure develops when the cell is placed in
glucose solution. What osmotic pressure develops when the cell is placed in 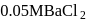 solution at
solution at 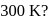
Detailed Solution: Question 14
A solution of 0.5 g of a solute (molar mass = 150 g mol−1) in 50 g of a solvent yields a boiling point elevation of 0.40 K. Another solution of 0.60 g of an unknown solute in the same mass of solvent exhibits a boiling point elevation of 0.8 K. The molar mass of unknown solute is :
Detailed Solution: Question 15
335 videos|699 docs|300 tests |


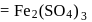
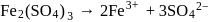 (complete ionization)
(complete ionization)
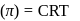
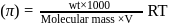
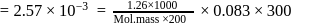
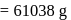
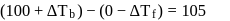
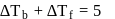
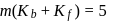
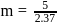 i.e.,
i.e., 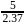 moles in
moles in 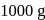 water
water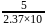 moles in
moles in 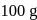 water
water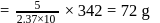
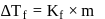
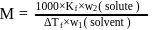
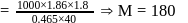
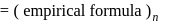
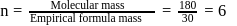
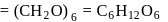
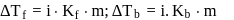
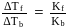
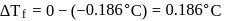
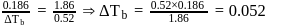
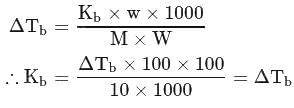
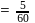 ;
;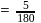
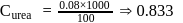
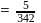
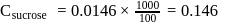
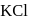 (effective)
(effective)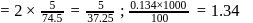
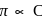
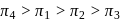
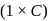 , higher the
, higher the 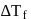 value and lower the freezing point.
value and lower the freezing point.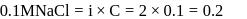
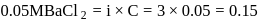
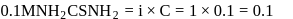
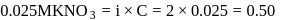
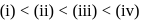
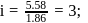
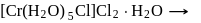
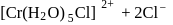
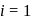
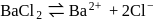
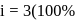 ionized
ionized 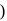
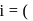 number of ions afterionization
number of ions afterionization