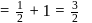Collision Theory, Energy of Activation - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Collision Theory, Energy of Activation (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Collision Theory, Energy of Activation". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
A first order reaction is half-completed in 45 minutes. How long does it need for  of the reaction to be completed?
of the reaction to be completed?
Detailed Solution: Question 1
At a certain temperature, the first order rate constant k1 is found to be smaller than the second order rate constant k2. If the energy of activation E1 of the first order reaction is greater than energy of activation E2 of the second order reaction, then with increase in temperature.
Detailed Solution: Question 2
A reaction which is of first order w.r.t. reactant  , has a rate constant
, has a rate constant  . If we start with
. If we start with 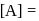
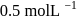 , when would [A] reach the value of
, when would [A] reach the value of 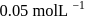
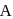 , has a rate constant
, has a rate constant 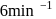 . If we start with
. If we start with 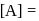
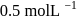 , when would [A] reach the value of
, when would [A] reach the value of 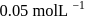
Detailed Solution: Question 3
The rate of a first order reaction is 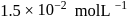
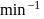 at
at  concentration of the reactant. The halflife of the reaction is
concentration of the reactant. The halflife of the reaction is
Detailed Solution: Question 4
For a first order reaction 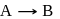 the reaction rate at reactant concentration of
the reaction rate at reactant concentration of 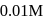 is found to be
is found to be 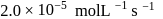 . The half life period of the reaction is
. The half life period of the reaction is
Detailed Solution: Question 5
A reaction proceeds by first order, 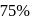 of this reaction was completed in 32 min. The time required for
of this reaction was completed in 32 min. The time required for 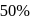 completion is
completion is
Detailed Solution: Question 6
In the reaction 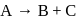 , rate constant is
, rate constant is 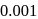
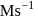 . If we start with
. If we start with 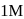 of A then conc. of
of A then conc. of 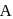 and B after 10 minuter are respectively.
and B after 10 minuter are respectively.
Detailed Solution: Question 7
The half life for the virus inactivation if in the beginning  of the virus is inactivated per minute is (Given: The reaction is of first order)
of the virus is inactivated per minute is (Given: The reaction is of first order)
Detailed Solution: Question 8
At 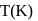 , if the rate constant of a first order reaction is
, if the rate constant of a first order reaction is 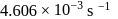 , the time to reduce the initial concentration of the reactant to
, the time to reduce the initial concentration of the reactant to 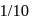 in seconds is :
in seconds is :
Detailed Solution: Question 9
The rate constant of a reaction with a virus is 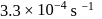 . Time required for the virus to become
. Time required for the virus to become 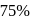 inactivated is
inactivated is
Detailed Solution: Question 10
A Geigger Muller counter is used to study the radioactive process. In the absence of radioactive substance  , it counts 3 disintegration per second (dps). At the start in the presence of
, it counts 3 disintegration per second (dps). At the start in the presence of  , it records 23 dps; and after 10 min 13 dps,
, it records 23 dps; and after 10 min 13 dps,
(i) What does it count after 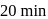
(ii) What is the halflife of A?
Detailed Solution: Question 11
The rate equation for a reaction,
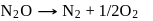
is Rate 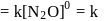 . If the initial concentration of the reactant is
. If the initial concentration of the reactant is 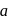 mol
mol 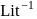 , the half-life period of the reaction is
, the half-life period of the reaction is
Detailed Solution: Question 12
Half-lives of first-order and zeroth order reactions are same. Ratio of rates at the start of nreaction is
Detailed Solution: Question 13
The plot of concentration of the reactant Vs time for a reaction is a straight line with a negative slope. The reaction follows a rate equation of
Detailed Solution: Question 14
The reaction 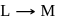 is started with
is started with 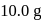 of L. After 30 and 90 minutes
of L. After 30 and 90 minutes 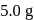 and
and 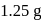 of
of  respectively are left. The order of the reaction is
respectively are left. The order of the reaction is
Detailed Solution: Question 15
The plot that represents the zero order reaction is:
Detailed Solution: Question 16
For a first order reaction, a plot of log(a−x) against time is a straight line with a negative slope equal to
Detailed Solution: Question 17
In the presence of an acid, the initial concentration of cane sugar was reduced from  to
to 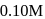 in 5 hours and from
in 5 hours and from  to
to  in 10 hours. The reaction is of :
in 10 hours. The reaction is of :
Detailed Solution: Question 18
For the decomposition of  at
at 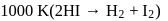 , the following data were obtained
, the following data were obtained
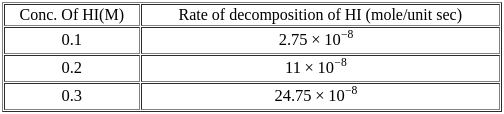
The order of reaction is :
 at
at 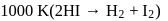 , the following data were obtained
, the following data were obtained
The order of reaction is :
Detailed Solution: Question 19
The hypothetical reaction 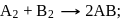 follows the following mechanism
follows the following mechanism  ,
,  The order of the overall reaction is
The order of the overall reaction is
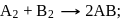 follows the following mechanism
follows the following mechanism  ,
,  The order of the overall reaction is
The order of the overall reaction isDetailed Solution: Question 20
335 videos|699 docs|300 tests |


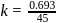 further
further
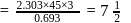 hours.
hours.
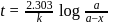
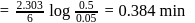
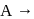 Products
Products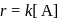 or
or 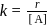
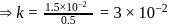
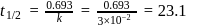
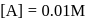
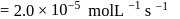
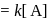

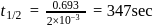
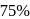 reaction gets completed in 32
reaction gets completed in 32 
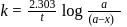
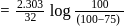

 to get the value of time required for
to get the value of time required for  completion of reaction
completion of reaction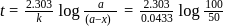
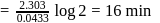
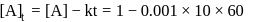

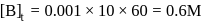


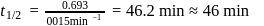
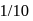 th, we use the formula:
th, we use the formula: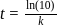
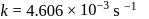 , we have:
, we have:
 , we get approximately 500 seconds.
, we get approximately 500 seconds.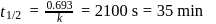
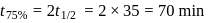
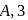 dps is zero error, hence
dps is zero error, hence 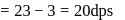
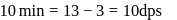
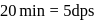
 recorded
recorded 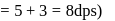
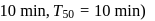
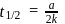
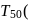 zeroth
zeroth 
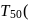 first
first 
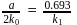
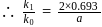

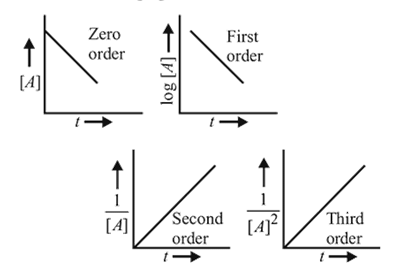
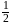 therefore
therefore 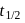 is 30 minutes. In 90 minutes the amount is reduced to
is 30 minutes. In 90 minutes the amount is reduced to 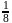 i.e.
i.e. 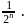 Here
Here 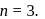 True for 1st order reaction.
True for 1st order reaction.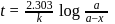
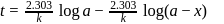
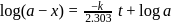
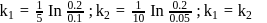
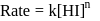
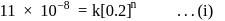

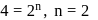
 ;
;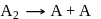 (Fast);
(Fast); (Slow)
(Slow)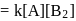 put value of
put value of 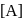 from Ist reaction since
from Ist reaction since 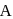 is intermediate
is intermediate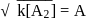
 Rate law equation
Rate law equation 
 Order
Order