Concentration of Solutions, Vapour pressure - Free MCQ Practice Test
MCQ Practice Test & Solutions: Test: Concentration of Solutions, Vapour pressure (15 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Concentration of Solutions, Vapour pressure". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
The molal elevation constant is the elevation in boiling point of
Detailed Solution: Question 1
Which of the following modes of expressing concentration is independent of temperature?
Detailed Solution: Question 2
How many grams of concentrated nitric acid solution should be used to prepare 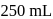 of
of  ? The concentrated acid is
? The concentrated acid is 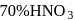
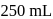 of
of  ? The concentrated acid is
? The concentrated acid is 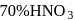
Detailed Solution: Question 3
Which of the following statements, regarding the mole fraction 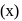 of a component in solution, is correct?
of a component in solution, is correct?
Detailed Solution: Question 4
The vapour pressure of water at 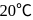 is 17.24 mm Hg. When 20 g of a non-ionic, substance is dissolved in 100 g of water, the vapour pressure is lowered by 0.30 mm Hg. What is the molecular mass of the substance?
is 17.24 mm Hg. When 20 g of a non-ionic, substance is dissolved in 100 g of water, the vapour pressure is lowered by 0.30 mm Hg. What is the molecular mass of the substance?
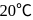 is 17.24 mm Hg. When 20 g of a non-ionic, substance is dissolved in 100 g of water, the vapour pressure is lowered by 0.30 mm Hg. What is the molecular mass of the substance?
is 17.24 mm Hg. When 20 g of a non-ionic, substance is dissolved in 100 g of water, the vapour pressure is lowered by 0.30 mm Hg. What is the molecular mass of the substance?Detailed Solution: Question 5
Two liquids X and Y form an ideal solution at 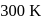 , vapour pressure of the solution containing 1 mol of X and 3 mol of Y is
, vapour pressure of the solution containing 1 mol of X and 3 mol of Y is 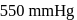 . At the same temperature, if 1 mol of Y is further added to this solution, vapour pressure of the solution increases by
. At the same temperature, if 1 mol of Y is further added to this solution, vapour pressure of the solution increases by 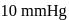 . Vapour pressure (in mmHg) of X and Y in their pure states will be, respectively–
. Vapour pressure (in mmHg) of X and Y in their pure states will be, respectively–
Detailed Solution: Question 6
Which of the following is not an ideal solution?
Detailed Solution: Question 7
An azeotropic solution of two liquids has boiling point lower than either of them when it
Detailed Solution: Question 8
Which among the following will show maximum osmotic pressure?
Detailed Solution: Question 9
Which of the following is not correct?
Detailed Solution: Question 10
A solution of acetone in ethanol
Detailed Solution: Question 11
An aqueous solution is 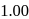 molal in KI. Which change will cause the vapour pressure of the solution to increase?
molal in KI. Which change will cause the vapour pressure of the solution to increase?
Detailed Solution: Question 12
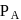 and
and 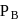 are the vapour pressure of pure liquid components,
are the vapour pressure of pure liquid components, 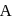 and
and 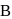 , respectively of an ideal binary solution. If
, respectively of an ideal binary solution. If 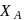 represents the mole fraction of component
represents the mole fraction of component 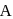 , the total pressure of the solution will be.
, the total pressure of the solution will be.
Detailed Solution: Question 13
Which condition is not satisfied by an ideal solution?
Detailed Solution: Question 14
Which of the following is true regarding azeotropes?
Detailed Solution: Question 15
335 videos|699 docs|300 tests |



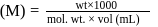
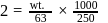
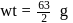
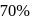 acid
acid 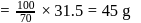
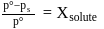
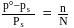

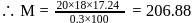
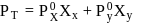
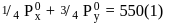
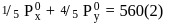
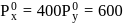
 Ideal solution
Ideal solution -dichlorobenzene
-dichlorobenzene  Ideal solution
Ideal solution Non-ideal solution
Non-ideal solution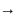 Ideal solution
Ideal solution in case of
in case of 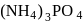 ,
,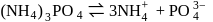
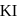 is diluted with water, concentration decreases, therefore the vapour pressure of the resulting solution increases.
is diluted with water, concentration decreases, therefore the vapour pressure of the resulting solution increases.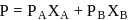
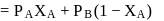
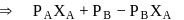
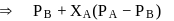
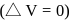 of mixing should be zero.
of mixing should be zero.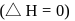 on mixing should be zero.
on mixing should be zero.