Concentration of the Ore - Free MCQ Practice Test with solutions, JEE Chemistry
MCQ Practice Test & Solutions: Test: Concentration of the Ore (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Concentration of the Ore". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following statements regarding metallurgy of iron is incorrect?
Detailed Solution: Question 1
Detailed Solution: Question 2
Which of the following condition favours the reduction of a metal oxide to metal?
Detailed Solution: Question 3
In order to refine "blister copper" it is melted in a furnace and is stirred with green logs of wood. The purpose is
Detailed Solution: Question 4
Match the items of List-I with those of List-II and choose the correct option given below.
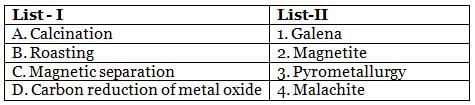

Detailed Solution: Question 5
Which of the following statements, about the advantage of roasting of sulphide ore before reduction is not true?
Detailed Solution: Question 6
Which one of the following is used to produce 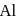 by electrolysis?
by electrolysis?
Detailed Solution: Question 7
Before introducing 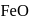 in blast furnace , it is converted to
in blast furnace , it is converted to 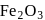 by roasting so that
by roasting so that
Detailed Solution: Question 8
Identify the correct statements from the following:
i. In the extraction of 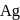 and
and 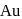 , zinc is used as reducing agent.
, zinc is used as reducing agent.
ii. Impure zinc can be refined by distillation method.
iii. Malachite is an ore of nickel.
i. In the extraction of
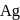 and
and 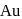 , zinc is used as reducing agent.
, zinc is used as reducing agent.ii. Impure zinc can be refined by distillation method.
iii. Malachite is an ore of nickel.
Detailed Solution: Question 9
Detailed Solution: Question 10
When a metal is to be extracted from its ore and the gangue associated with the ore is silica, then
Detailed Solution: Question 11
Which of the following statements is not correct?
1. Reduction of alumina to give aluminium by magnesium is thermodynamically feasible.
2. The point of intersection of 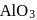 and
and 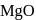 curves in Ellingham diagram is below
curves in Ellingham diagram is below 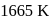 .
.
3. Use of magnesium as reducing agent in metallurgy of aluminium is economical.
4. Ellingham diagram represents the graphical plot of Gibbs energy vs temperature for the formation of the oxides of common metals and reducing agents.
Detailed Solution: Question 12
Which of the following examples is not correctly matched?
Detailed Solution: Question 13
Froth floatation process is used for the metallurgy of
Detailed Solution: Question 14
Which of the following metal is leached by cyanide process
Detailed Solution: Question 15
Sulphide ores of metals are usually concentrated by froth flotation process. Which one of the following sulphide ores offer an exception and is concentrated by chemical leaching?
Detailed Solution: Question 16
One of the processes used for concentration of ores is Froth floatation process. This process is generally used for concentration of sulphide ores. Sometimes in this process we add 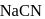 as a depressant.
as a depressant. 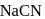 is generally added in case of
is generally added in case of 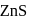 and PbS minerals. What is the purpose of addition of NaCN during the process of Froth floatation ?
and PbS minerals. What is the purpose of addition of NaCN during the process of Froth floatation ?
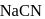 as a depressant.
as a depressant. 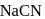 is generally added in case of
is generally added in case of 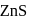 and PbS minerals. What is the purpose of addition of NaCN during the process of Froth floatation ?
and PbS minerals. What is the purpose of addition of NaCN during the process of Froth floatation ?Detailed Solution: Question 17
Which of the following statements is not correct?
(i) Froth-floatation is used for removing gangue from sulphide ore.
(ii) Cresols are used to stabilise the froth.
(iii) Sodium cyanide can be used as depressant for preferential separation.
(iv) Aniline can be used as froth enhancer.
Detailed Solution: Question 18
Which of the following reactions is an example for calcination process?
Detailed Solution: Question 19
Which one of the following statements is true about the froth flotation method?
Detailed Solution: Question 20
335 videos|699 docs|300 tests |


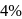 carbon and impurities like
carbon and impurities like 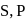 Mn etc., in small amount.
Mn etc., in small amount.
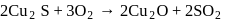
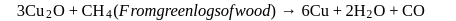
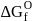 of most of the sulphides are greater than those of
of most of the sulphides are greater than those of 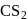 and
and 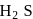 , therefore neither
, therefore neither 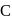 nor
nor 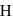 can reduce metal sulphide to metal. Further, the standard free energies of formation of oxide are much less than those of
can reduce metal sulphide to metal. Further, the standard free energies of formation of oxide are much less than those of 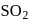 . Hence oxidation of metal sulphides to metal oxide is thermodynamically favourable.
. Hence oxidation of metal sulphides to metal oxide is thermodynamically favourable.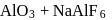 as elecurolyse, carbon
as elecurolyse, carbon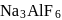 or
or 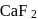 (cryolite or fluorspar), which lowers the melting point of alumina and brings conductivity.
(cryolite or fluorspar), which lowers the melting point of alumina and brings conductivity.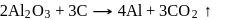
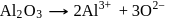
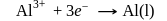 At anode
At anode 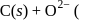 melt
melt 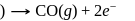
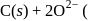 melt
melt 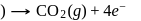
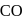 and
and 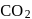 . Hence, option (a) is the correct answer.
. Hence, option (a) is the correct answer.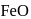 is capable forming slag with
is capable forming slag with 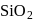
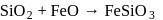
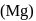 as reducing agent in metallurgy of aluminium
as reducing agent in metallurgy of aluminium 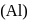 is uneconomical. So, statement 3 is not correct.
is uneconomical. So, statement 3 is not correct.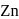 and
and 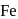 occur only in combined state, as these metals are available in nature mostly in the form of sulphide and oxides respectively.
occur only in combined state, as these metals are available in nature mostly in the form of sulphide and oxides respectively.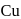 and
and 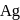 occur in both native and combined state.
occur in both native and combined state.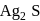 is an ore of silver. Silver is extracted from argentite by the mac-Arthur and Forest process (leaching process).
is an ore of silver. Silver is extracted from argentite by the mac-Arthur and Forest process (leaching process).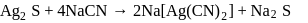
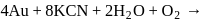
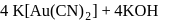
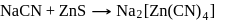
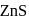 and due to this
and due to this 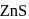 is prevented from the froth formation while PbS form froth.
is prevented from the froth formation while PbS form froth.