Covalent and Co-ordinate Bonding - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Covalent and Co-ordinate Bonding (15 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Covalent and Co-ordinate Bonding". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
Among the following ions, the pπ−dπ overlap could be present in
Detailed Solution: Question 1
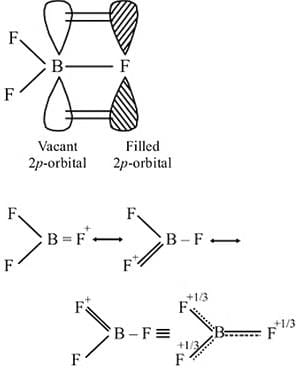
The bond dissociation energy of B−F in BF3 is 646 kJ mol−1 whereas that of C−F in CF4 is 515 kJ mol−1. The correct reason for higher B−F bond dissociation energy as compared to that of C−F is
Detailed Solution: Question 2
In 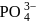 ion the formal charge on the oxygen atom of P−O bond is ___
ion the formal charge on the oxygen atom of P−O bond is ___
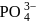 ion the formal charge on the oxygen atom of P−O bond is ___
ion the formal charge on the oxygen atom of P−O bond is ___Detailed Solution: Question 3
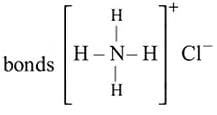
Which of the following contains both covalent and ionic bond?
Detailed Solution: Question 4
Which of the following species is a radical?
Detailed Solution: Question 5
The correct order in which the O − O bond length increases in the following is-
Detailed Solution: Question 6
Which of the following Lewis dot structure of  is incorrect?
is incorrect?
Detailed Solution: Question 7
The molecules  and
and 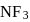 are both covalent compounds, but
are both covalent compounds, but 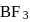 is non polar whereas
is non polar whereas 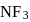 is polar. The reason for this is
is polar. The reason for this is
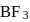 and
and 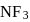 are both covalent compounds, but
are both covalent compounds, but 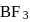 is non polar whereas
is non polar whereas 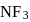 is polar. The reason for this is
is polar. The reason for this isDetailed Solution: Question 8
The number and type of bonds in ion in 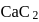 are:
are:
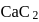 are:
are:Detailed Solution: Question 9
In 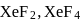 and
and 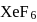 , the number of lone pairs on Xe are respectively
, the number of lone pairs on Xe are respectively
Detailed Solution: Question 10
The number of 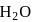 molecules participating in hydrogen bonding in
molecules participating in hydrogen bonding in 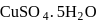 is/are
is/are
Detailed Solution: Question 11
The maximum number of H-bonds in which water molecule can participate is
Detailed Solution: Question 12
Which of the following molecules has highest dipole moment?
Detailed Solution: Question 13
Which of the following has least covalent P − H bond?
Detailed Solution: Question 14
Which one of the following has longest covalent bond distance?
Detailed Solution: Question 15
332 videos|699 docs|300 tests |



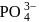 ion, formal charge on each
ion, formal charge on each  -atom of
-atom of 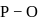
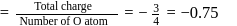
 Odd electrons (seven valency electrons of nitrogen and eight valency electrons of oxygen), so this species work as radical.
Odd electrons (seven valency electrons of nitrogen and eight valency electrons of oxygen), so this species work as radical. 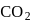 , the total electrons are
, the total electrons are 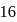 (even), in case of nitro ion, total valency electrons are
(even), in case of nitro ion, total valency electrons are 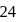 (even), In case of cyanide ion, total valency electrons are
(even), In case of cyanide ion, total valency electrons are 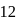 (even).
(even).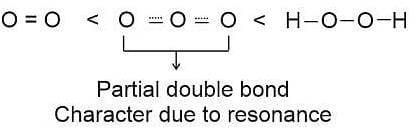
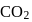 can be represented as
can be represented as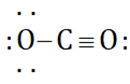
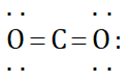 (Incorrect structure)
(Incorrect structure)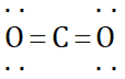
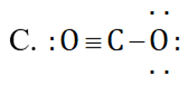
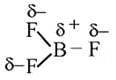
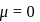 hence it is non polar.
hence it is non polar.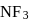 is pyramidal
is pyramidal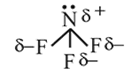
 hence it is polar.
hence it is polar. is
is 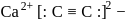
 and two
and two  bonds
bonds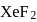 Total number of valence electrons of
Total number of valence electrons of 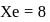 , two electrons shared with
, two electrons shared with 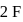 atoms, 6 electrons left hence 3 lone pairs, in
atoms, 6 electrons left hence 3 lone pairs, in 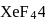 shared with
shared with 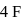 atoms 4 left hence 2 lone pairs; in
atoms 4 left hence 2 lone pairs; in 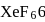 shared with
shared with 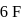 atoms 2 left hence 1 lone pair.
atoms 2 left hence 1 lone pair.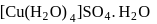 is the represenation of given compound.
is the represenation of given compound.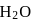 molecule, two H-bonds are formed with
molecule, two H-bonds are formed with 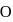 atom and two are formed with
atom and two are formed with 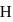 atoms of the
atoms of the 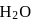 molecule.
molecule.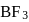 and
and 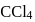 is zero because these are symmetrical molecules. Dipole moment of
is zero because these are symmetrical molecules. Dipole moment of 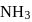 is greater than
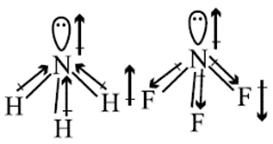
is greater than 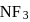 .
.
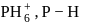 bond will have ionic character due to electron attracting tendency of P-carrying positive charge and least covalent.
bond will have ionic character due to electron attracting tendency of P-carrying positive charge and least covalent.