Determination of Rate a Reaction, Arrhenius theory - Free MCQ Practice
MCQ Practice Test & Solutions: Test: Determination of Rate of a Reaction, Arrhenius theory (15 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Determination of Rate of a Reaction, Arrhenius theory". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
The time taken for 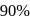 of a first order reaction to complete is approximately
of a first order reaction to complete is approximately
Detailed Solution: Question 1
A foreign substance that increase the speed of a chemical reaction is called
Detailed Solution: Question 2
. What will be the value of instantaneous rate of reaction from the graph?
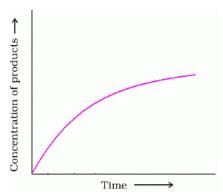

Detailed Solution: Question 3
The rate of a chemical reaction doubles for every 10°C rise of temperature. If the temperature is raised by 50°C, the rate of the reaction increases by about
[AIEEE 2011]
Detailed Solution: Question 4
Chemical substances speeding up rate of chemical reaction is called as
Detailed Solution: Question 5
Which of the following reactions is not of the first order?
Detailed Solution: Question 6
A first order reaction is 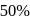 completed in 20 minutes at
completed in 20 minutes at 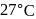 and in 5 minutes at
and in 5 minutes at 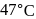 . The energy of activation of the reaction is :
. The energy of activation of the reaction is :
Detailed Solution: Question 7
In a first-order reaction 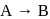 , if
, if 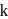 is rate constant and initial concentration of the reactant
is rate constant and initial concentration of the reactant 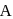 is
is 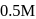 , then the halflife is
, then the halflife is
Detailed Solution: Question 8
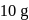 of a radioactive element is disintegrated to
of a radioactive element is disintegrated to  in 2.303 minutes. What is the half-life (in minutes) of that radioactive element?
in 2.303 minutes. What is the half-life (in minutes) of that radioactive element?
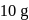 of a radioactive element is disintegrated to
of a radioactive element is disintegrated to 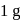 in 2.303 minutes. What is the half-life (in minutes) of that radioactive element?
in 2.303 minutes. What is the half-life (in minutes) of that radioactive element?Detailed Solution: Question 9
Half life period of a first-order reaction is 1386 seconds. The specific rate constant of the reaction is:
Detailed Solution: Question 10
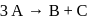 , it would be a zero order reaction when
, it would be a zero order reaction when
Detailed Solution: Question 11
The decomposition of a substance follows first order kinetics. Its concentration is reduced to 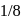 th of its initial value in 24 minutes. The rate constant of the decomposition process in
th of its initial value in 24 minutes. The rate constant of the decomposition process in 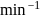 is
is
Detailed Solution: Question 12
The time required for completion of 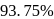 of a first order reaction is
of a first order reaction is 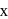 minutes. The half life of it (in minutes) is
minutes. The half life of it (in minutes) is
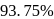 of a first order reaction is
of a first order reaction is 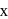 minutes. The half life of it (in minutes) is
minutes. The half life of it (in minutes) isDetailed Solution: Question 13
The halflife of a radioactive element is 20 min. The time interval between the stages of its 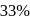 and
and 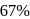 decay is
decay is
Detailed Solution: Question 14
The reaction 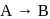 follows first order kinetics. The time taken for
follows first order kinetics. The time taken for 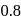 mole of
mole of 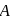 to produce
to produce 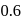 mole of
mole of 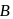 is 1 hour. What is the time taken for conversion of
is 1 hour. What is the time taken for conversion of 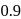 mole of
mole of 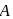 to produce
to produce 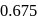 mole of B?
mole of B?
Detailed Solution: Question 15
335 videos|699 docs|300 tests |


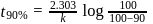 (I)
(I)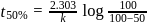 (II)
(II)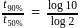
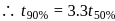

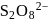 and
and  .
.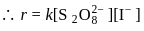
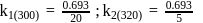
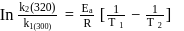
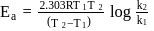
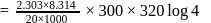
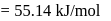
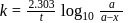
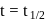
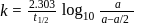
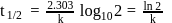
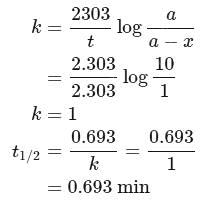
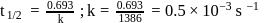
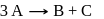
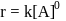 , i. e the rate remains same at any concentration of '
, i. e the rate remains same at any concentration of ' 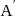 . i.e independent upon concentration of A.
. i.e independent upon concentration of A.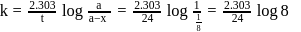
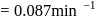

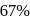 to
to 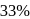 is almost half the concentration change.
is almost half the concentration change.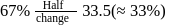
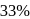 and
and 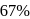 decay is same as
decay is same as 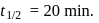
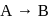 For a first order reaction
For a first order reaction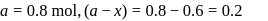
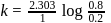 or
or 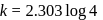
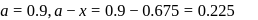
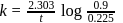
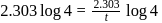
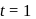 hour
hour