Different Methods of Expressing Concentration Solutions - Free MCQ Practice
MCQ Practice Test & Solutions: Test: Different Methods of Expressing Concentration of Solutions (30 Questions)
You can prepare effectively for JEE Chapter-wise Tests for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Different Methods of Expressing Concentration of Solutions". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 45 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Only One Option Correct Type
This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct
Which of the following concentration factors is affected by change in temperature?
This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct
Detailed Solution: Question 1
6.02 x1020 molecules of urea are present in 100 mL of its solution. The concentration of urea solution is (N0 = 6.02 x 1023 mol-1)
[AIEEE 2004]
Detailed Solution: Question 2
Glucose solution is one molal. Glucose present in 1 kg glucose solution is
Detailed Solution: Question 3
The molality of a urea solution in which 0.0100 g of urea (NH2CONH2) is added to 0.300 dm3 of water at STP is
[AIEEE 2011]
Detailed Solution: Question 4
A solution that is 20% ethanol by volume is found to have a density of 0.977 g/mL. Density of ethanol is 0.789 g/mL. Thus, mass per cent of ethanol solution is
Detailed Solution: Question 5
A solution of NH4CI is prepared by dissolving 95 g of NH4CI in 200 g of H2O at 60° C. What is mass per cent when the solution is cooled to 20° C based on this
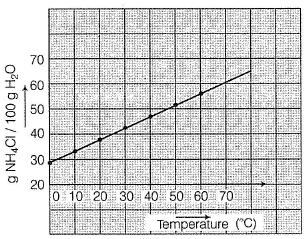
Detailed Solution: Question 6
Commercial concentrated nitric acid is 15.6 M. To prepare 10 L of 6.0 M nitric acid from it,
Detailed Solution: Question 7
Mole fraction of C3H5(OH)3 in a solution of 36 gm of water and 46 gm of glycerine is
Detailed Solution: Question 8
AgNO3 sample is 85% by mass. To prepare 125 mL of 0.05 M AgNO3 solution, AgNO3 sample required is
Detailed Solution: Question 9
Hardness of a water sample is 200 ppm CaCO3, Thus, molarity of CaCO3 is
Detailed Solution: Question 10
100 mL of aqueous solution of 0.01 M CaCI2 is evaporated to dryness when 0.15 g of residue is obtained. Thus, impurity present is
Detailed Solution: Question 11
At 25 ° C, the density of 18 MH2SO4 is 1.8 g cm3. Thus, mass percentage of H2SO4 in aqueous solution is
Detailed Solution: Question 12
Laboratory ammonia is 14.8 M NH3(ag)with a density of 0.8980 g/mL. Mole fraction of NH3 in this solution is
Detailed Solution: Question 13
An aqueous solution is 34% H3P04 by mass and has density 1.209 g mL -1 Molarity (I), molality (II) and normality (III) respectively, are
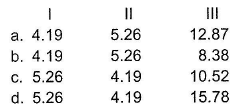
Detailed Solution: Question 14
If one assumes the volumes are additive, what is 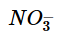 in a solution obtained by mixing 275 mL of 0.200 M KN03, 325 mL of 0.40 M Mg(N03)2 and 400 mL H20?
in a solution obtained by mixing 275 mL of 0.200 M KN03, 325 mL of 0.40 M Mg(N03)2 and 400 mL H20?
Detailed Solution: Question 15
How many grams of water would you add to 1.38 moles of CH3OH in 1 kg water to reduce the molality to 1.00 molal CH3OH(aq) ?
Detailed Solution: Question 16
Three solutions have been provided.
I. 3 g HCI in 1 kg H20.
II. 2 L HCI(g) and 1 L H20 at room temperature.
III. Aqueous solution of HCI with 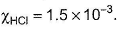
Solutions containing same number of moles of HCI is/are
II. 2 L HCI(g) and 1 L H20 at room temperature.
III. Aqueous solution of HCI with
Detailed Solution: Question 17
Which of the following aqueous solutions has the highest concentration of Na+?
Detailed Solution: Question 18
pH of Ba(OH)2 solution is 12. Number of millimoles present in 100 mL of Ba(OH)2 solution is
Detailed Solution: Question 19
We have
I. 25 mL of 1 M NaOH
II. 10 mL of 0.50 M NaCI
On mixing the two solutions, molar concentrations of Na+, OH- and Cl- respectively, are

I. 25 mL of 1 M NaOH
II. 10 mL of 0.50 M NaCI
On mixing the two solutions, molar concentrations of Na+, OH- and Cl- respectively, are
Detailed Solution: Question 20
A ‘100 proof solution of ethanol in water consists of 50.0 mL of C2H5OH(/)and 50.0 mL of H20 (/), mixed at 16 °C . Given,
Density of H20 = 1 g mL-1
Density of C2H,OH = 0.7939 gmL-1
Density mixture = 0.9344 g mL-1
Volume of the solution is
Density of H20 = 1 g mL-1
Density of C2H,OH = 0.7939 gmL-1
Density mixture = 0.9344 g mL-1
Volume of the solution is
Detailed Solution: Question 21
How many grams of Mgl2 must be added to 250 mL of 0.0876 M Kl to produce a solution with [l-] = 0.10 M?
Detailed Solution: Question 22
An aqueous solution is 6% methanol, CH3OH, by mass with d = 0.988 g mL-1. Thus, molarity of CH3OH in this solution is
Detailed Solution: Question 23
Matching List Type
Choices for the correct combination of elements from Column I and Column II are given as options (a), (b), (c) and (d), out of which one is correct
An ethanol-water solution is prepared by dissolving 10.00 mL o f ethanol (C2H5OH, d = 0.789 g mL-1) in a sufficient volume o f w ater to produce 100 mL of solution with a density, d = 0.982 g mL-1. Match the concentration term in Column I with its value in Column II and select the answer from the code
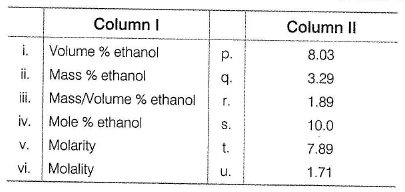

Choices for the correct combination of elements from Column I and Column II are given as options (a), (b), (c) and (d), out of which one is correct
Detailed Solution: Question 24
One or More than One Options Correct Type
This section contains 2 multiple type questions. Each question has 4 choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
1 kg of aqueous solution has 0.40 kg NaOH. Thus, this solution is
This section contains 2 multiple type questions. Each question has 4 choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
1 kg of aqueous solution has 0.40 kg NaOH. Thus, this solution is
Detailed Solution: Question 25
Mole fraction of ethanol in ethanol-water solution is 0.25. Thus, this solution is
Detailed Solution: Question 26
Comprehension Type
This section contains a passage describing theory, experiments, data, etc. Two questions related to the paragraph have been given. Each question has only one correct answer out of the given 4 options (a), (b), (c) and (d)
Passage
A handbook gives the concentration of water in a 2.772 M aqueous solution of NaOH as 998.0 g L-1.
Q. Molality of solution is
Detailed Solution: Question 27
Comprehension Type
This section contains a passage describing theory, experiments, data, etc. Two questions related to the paragraph have been given. Each question has only one correct answer out of the given 4 options (a), (b), (c) and (d)
Passage
A handbook gives the concentration of water in a 2.772 M aqueous solution of NaOH as 998.0 g L -1.
Q.
Molality of solution is
Detailed Solution: Question 28
One Integer Value Correct Type
This section contains 1 questions, when worked out will result in an integer value from 0 to 9 (both inclusive)
Q. Certain brine solution has 3.87% NaCI by mass with density 1.036 g mL-1. How many millilitre of this solution should be evaporated to obtain 0.32 g of NaCI?
This section contains 1 questions, when worked out will result in an integer value from 0 to 9 (both inclusive)
Detailed Solution: Question 29
A 5.2 molal aqueous solution of methyl alcohol (CH3OH) is supplied. What is the mole fraction of methyl alcohol in the solution?
Detailed Solution: Question 30
446 docs|929 tests |



