Electromagnetic radiation, Bohr model - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Electromagnetic radiation, Bohr model (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Electromagnetic radiation, Bohr model". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
What is the angular velocity 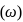 of an electron occupying second orbit of
of an electron occupying second orbit of 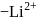 ion?
ion?
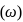 of an electron occupying second orbit of
of an electron occupying second orbit of 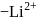 ion?
ion?Detailed Solution: Question 1
The ion that is isoelectronic with CO is-
Detailed Solution: Question 2
The energy of an electron in first Bohr orbit of 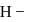 atom is
atom is 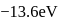 . The energy value of electron in the excited state of
. The energy value of electron in the excited state of 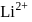 is:
is:
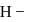 atom is
atom is 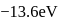 . The energy value of electron in the excited state of
. The energy value of electron in the excited state of 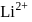 is:
is:Detailed Solution: Question 3
The Bohr's energy equation for H atom reveals that the energy level of a shell is given by E = −13.58/n2eV. The smallest amount that an H atom will absorb if in ground state is
Detailed Solution: Question 4
If the electron of a hydrogen atom is present in the first orbit, the total energy of the electron is
Detailed Solution: Question 5
An electron, e1 is moving in the fifth stationary state, and another electron e2 is moving in the fourth stationary state. The radius of orbit of electron, e1 is five times the radius of orbit of electron, e2 calculate the ratio of velocity of electron e1(v1) to the velocity of electron e2(v2)
Detailed Solution: Question 6
In a hydrogen atom, if energy of an electron 1n ground state is 13.6.ev, then that in the 2nd excited state is
Detailed Solution: Question 7
What is the ratio of the atomic radius of the 5th orbit in chlorine atom and 3rd orbit in Helium atom?
Detailed Solution: Question 8
An electron in an atom jumps in such a way that its kinetic energy changes from 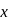 to
to 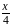 . The change in potential energy will be:
. The change in potential energy will be:
Detailed Solution: Question 9
What atomic number of an element "X" would have to become so that the 4 th orbit around X would fit inside the I Bohr orbit of H atom?
Detailed Solution: Question 10
If in Bohr's model, for unielectronic atom, time period of revolution is represented as 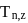 where n represents shell no. and
where n represents shell no. and 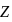 represents atomic number then the value of
represents atomic number then the value of 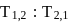 will be :
will be :
Detailed Solution: Question 11
The energy of 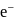 in first orbit of
in first orbit of 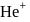 is
is 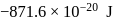 . The energy of
. The energy of 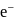 in first orbit of
in first orbit of 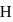 is:
is:
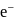 in first orbit of
in first orbit of 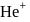 is
is 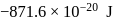 . The energy of
. The energy of  in first orbit of
in first orbit of 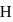 is:
is:Detailed Solution: Question 12
If radius of second stationary orbit (in Bohr's atom) is 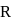 then radius of third orbit will be:
then radius of third orbit will be:
Detailed Solution: Question 13
Detailed Solution: Question 14
The energy of an electron in first Bohr orbit of 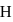 -atom is
-atom is 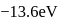 . The possible energy value of electron in the excited state of
. The possible energy value of electron in the excited state of 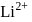 is
is
Detailed Solution: Question 15
The kinetic energy of an electron in the second Bohr orbit of a hydrogen atom is [a0 is Bohr radius] :
Detailed Solution: Question 16
The potential energy of the electron present in the ground state of 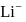 ion is represented by:
ion is represented by:
Detailed Solution: Question 17
The ratio of kinetic energy and total energy of an electron in Bohr orbit of hydrogen-like species is
Detailed Solution: Question 18
The speed of an electron in the first Bohr orbit is 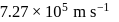 . The speed of electron in the
. The speed of electron in the 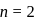 level of
level of 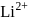 ion will be
ion will be
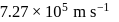 . The speed of electron in the
. The speed of electron in the 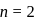 level of
level of 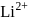 ion will be
ion will beDetailed Solution: Question 19
Which of the following statements regarding Bohr theory of hydrogen atom is not correct?
Detailed Solution: Question 20
335 videos|697 docs|300 tests |


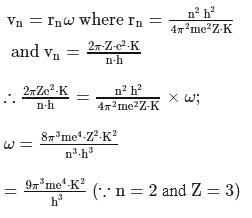

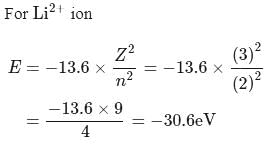
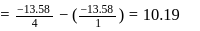
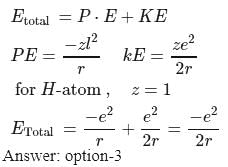
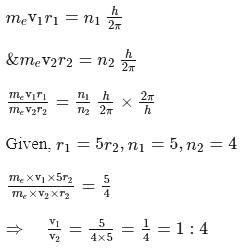
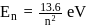 or
or 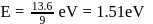
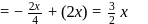
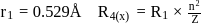
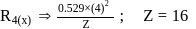
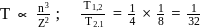

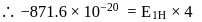
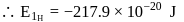
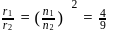
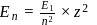
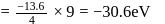
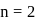 and for
and for 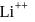 ion,
ion, 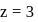
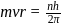
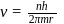
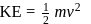
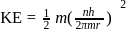
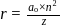
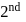 Bohr orbit
Bohr orbit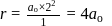
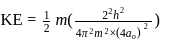
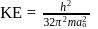
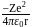
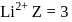 , where
, where 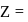 atomic number of an atom
atomic number of an atom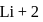 ion is
ion is 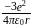
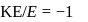
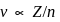 , we will have
, we will have 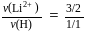
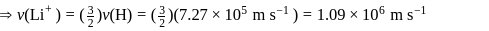
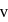 around the nucleus in a stationary orbit of radius
around the nucleus in a stationary orbit of radius 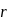 . The electrostatic force acting on the electron is balanced by the centrifugal force i.e.
. The electrostatic force acting on the electron is balanced by the centrifugal force i.e. 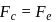
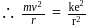 where
where 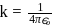
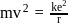 ... (1)
... (1)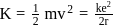 ... (2)
... (2)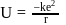
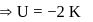 (using (2))
(using (2))