General Properties of Group-16 - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: General Properties of Group-16 (14 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: General Properties of Group-16". These 14 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 14
Sign up on EduRev for free to attempt this test and track your preparation progress.
Non-metals combine with oxygen to form usually
Detailed Solution: Question 1
Detailed Solution: Question 2
Detailed Solution: Question 3
Which one of the following statements is not correct?
Detailed Solution: Question 4
Tailing of mercury test can be used for which of the following gas?
Detailed Solution: Question 5
By which of the following methods, 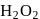 can't synthesized?
can't synthesized?
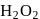 can't synthesized?
can't synthesized?Detailed Solution: Question 6
A green coloured solution of same salt changes its colour to light pink on passing ozone through it. Which of the following species represent pink and green colour respectively?
Detailed Solution: Question 7
Which of the following on thermal decomposition gives oxygen gas ?
Detailed Solution: Question 8
Correct order of 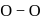 bond length (increasing) in
bond length (increasing) in 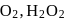 and
and 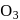 is
is
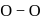 bond length (increasing) in
bond length (increasing) in 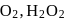 and
and 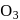 is
isDetailed Solution: Question 9
Which of the following statements regarding ozone is not correct?
Detailed Solution: Question 10
Which of the following oxidation state of oxygen is not observed in its compounds
Detailed Solution: Question 11
An organic compound containing sulphur is estimated by Carius method in which fuming 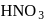 is used to convert
is used to convert 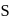 into :
into :
Detailed Solution: Question 12
Identify the correct statements from the following.
(i) Oxygen shows 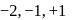 and +2 oxidation states.
and +2 oxidation states.
(ii) The thermal stability of 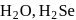 and
and 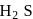 follows the order
follows the order 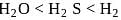 Se.
Se.
(iii) The reducing nature of 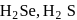 and
and 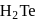 follows the order
follows the order 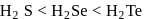
(i) Oxygen shows
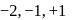 and +2 oxidation states.
and +2 oxidation states.(ii) The thermal stability of
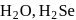 and
and 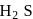 follows the order
follows the order 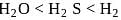 Se.
Se.(iii) The reducing nature of
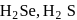 and
and 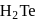 follows the order
follows the order 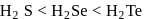
Detailed Solution: Question 13
Photochemical smog consists of excessive amount of 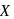 , in addition to aldehydes, ketones, peroxy-acetyl nitrile (PAN), and so forth,
, in addition to aldehydes, ketones, peroxy-acetyl nitrile (PAN), and so forth, 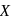 is:
is:
Detailed Solution: Question 14
335 videos|697 docs|300 tests |


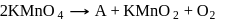 is
is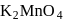

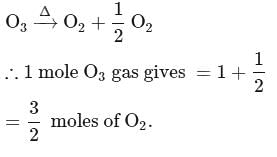
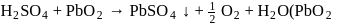 is not peroxy compound)
is not peroxy compound)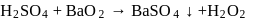
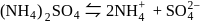
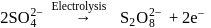
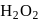 .
.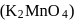 having manganate ion
having manganate ion 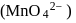 .
.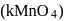 having permangate ion
having permangate ion 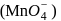 ,
,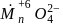 (green in colour) to
(green in colour) to 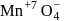 (Light pink in colour).
(Light pink in colour).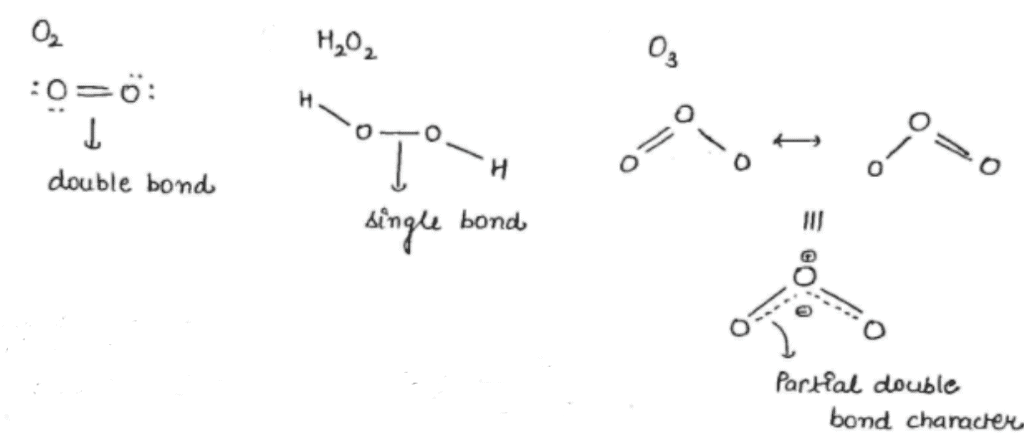
 < Single bond
< Single bond and double
and double 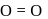 bonds. The bond angle is
bonds. The bond angle is 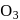 is
is 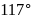 with
with 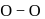 distance
distance 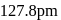 .
.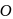 do not show
do not show 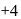 oxidation state because its valency is only
oxidation state because its valency is only 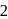 .
. 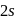 electrons takes more energy to ionize.
electrons takes more energy to ionize.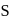 is converted to
is converted to 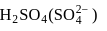 .
.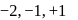 and +2 oxidation states.
and +2 oxidation states.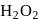 ), +1 in
), +1 in 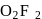 and +2 in
and +2 in 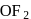 .
.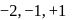 and +2 oxidation states.
and +2 oxidation states.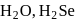 and
and 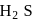 follows the order
follows the order 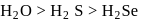 This is because, on going down the group, the size of the central atom increases and therefore, its tendency to form stable covalent bond with hydrogen decreases. As a result, the
This is because, on going down the group, the size of the central atom increases and therefore, its tendency to form stable covalent bond with hydrogen decreases. As a result, the 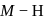 bond strength decreases and therefore, thermal stability decreases.
bond strength decreases and therefore, thermal stability decreases.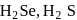 and
and 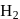 Te follows the order
Te follows the order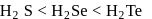
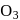 .
.