Grignard Reagent - Free MCQ Practice Test with solutions, Chemistry Organic
MCQ Practice Test & Solutions: Test: Grignard Reagent (10 Questions)
You can prepare effectively for Chemistry Organic Chemistry with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Grignard Reagent". These 10 questions have been designed by the experts with the latest curriculum of Chemistry 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 30 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following reaction sequence that will best carry out the following preparation?

Detailed Solution: Question 1
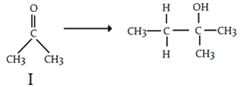
Which of the following reagents, when treated with phenylmagnesiuim bromide followed by acid workup, will yield 2-phenylethanol?
Detailed Solution: Question 2
A Grignard’s reagent may be made by reacting magnesium with which of the following compound?
Detailed Solution: Question 3
Which of the following statements about Grignard reagent is false?
Detailed Solution: Question 4
Detailed Solution: Question 5
Which of the following compounds would not give tert-butyl alcohol when treated with excess methylmagnesium bromide?
Detailed Solution: Question 6
Which of the following compounds gives a primary alcohol upon reaction with phenylmagnesium bromide?
Detailed Solution: Question 7
Which of the following compounds gives a secondary alcohol upon reaction with methylmagnesium bromide?
Detailed Solution: Question 8
Which of the following compounds does not give a tertiary alcohol upon reaction with methylmagnesium bromide/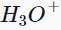 ?
?
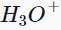 ?
?Detailed Solution: Question 9
Alkyl halides can be converted into Grignard reagents by _______________
Detailed Solution: Question 10
39 videos|96 docs|46 tests |