Group 17 Elements: Halogens - Free MCQ Practice Test with solutions, JEE
MCQ Practice Test & Solutions: Test: Group 17 Elements: Halogens (15 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Group 17 Elements: Halogens". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
A white precipitate is obtained on hydrolysis of
Detailed Solution: Question 1
Which of the following arrangements gives the correct order of increasing oxidation number of iodine?
Detailed Solution: Question 2
Mark the element which shows only one oxdation state
Detailed Solution: Question 3
Which of the following is not correctly matched?
Detailed Solution: Question 4
Detailed Solution: Question 5
What are the products formed when chlorine is passed through aqueous hypo solution?
Detailed Solution: Question 6
Which one of the following reactions does not occur?
Detailed Solution: Question 7
Of the interhalogen 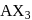 compounds,
compounds, 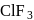 is most reactive but
is most reactive but 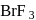 has higher conductance
has higher conductance
in liquid state. This is because
Detailed Solution: Question 8
Which of the following is the best description for the behaviour of bromine in the reaction given below ? 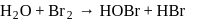
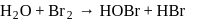
Detailed Solution: Question 9
Detailed Solution: Question 10
Which of the following is correct about the reaction?
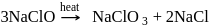
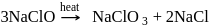
Detailed Solution: Question 11
Detailed Solution: Question 12
The correct order of reactivity of halogens with alkalies is
Detailed Solution: Question 13
Among the following oxoacids, the correct order of acid strength is
Detailed Solution: Question 14
Which of the following species is not a pseudo halide :-
Detailed Solution: Question 15
332 videos|699 docs|300 tests |


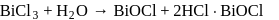

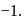
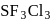 is
is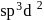 , hence octahedral
, hence octahedral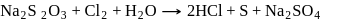
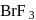 dissociates into
dissociates into 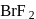 and
and 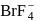 ions most easily.
ions most easily.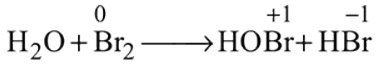
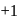 and also decreases from 0 to
and also decreases from 0 to 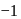 . Thus it is oxidised as well as reduced.
. Thus it is oxidised as well as reduced.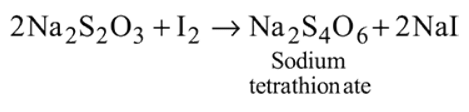
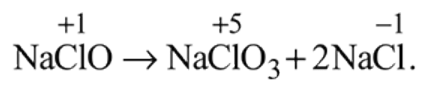
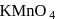 and
and 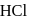 results in
results in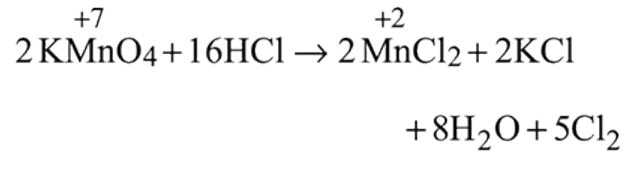
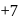 to
to 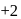 hence reduction occurs and
hence reduction occurs and 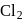 is formed.
is formed.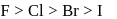
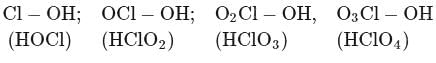
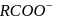 is not a pseudohlide ion. Remaining
is not a pseudohlide ion. Remaining 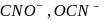 and
and 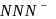 are pseudohlide ions.
are pseudohlide ions.