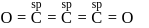Hybrididation and VSEPR theory - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Hybrididation and VSEPR theory (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Hybrididation and VSEPR theory". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
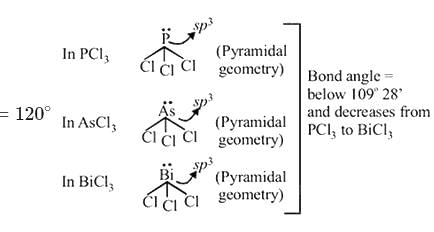
In compounds of type ECl3, where E = B,P, As or Bi, the angles Cl − E − Cl for different E are in the order.
Detailed Solution: Question 1
For which of the following sets of geometry, both axial and equatorial positions are present?
Detailed Solution: Question 2
Detailed Solution: Question 3
Which one of the following is a correct pair with respect to molecular formula of xenon compound and hybridization state of xenon in it?
Detailed Solution: Question 4
Chlorine atom, in its third excited state, reacts with fluorine to form a compound  . The formula and shape of
. The formula and shape of  are :
are :
Detailed Solution: Question 5
The bond angle between two hybrid orbitals is 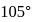 Calculate the percentage of
Calculate the percentage of 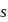 -character of hybrid orbital.
-character of hybrid orbital.
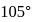 Calculate the percentage of
Calculate the percentage of 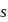 -character of hybrid orbital.
-character of hybrid orbital.Detailed Solution: Question 6
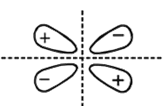
Main axis of a diatomic molecule is Z. AO's 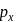 and
and 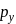 overlap to form which of the following orbitals?
overlap to form which of the following orbitals?
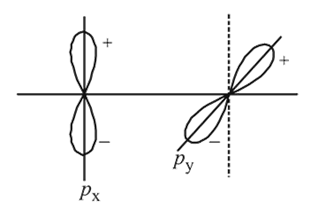
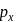 and
and 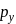 overlap to form which of the following orbitals?
overlap to form which of the following orbitals?
Detailed Solution: Question 7
Observe the following statements:
i. According to VSEPR theory, 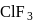 and
and 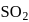 are shown
are shown 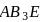 and
and 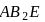 type molecules respectively.
type molecules respectively.
ii. 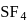 has "See-saw" shape.
has "See-saw" shape.
iii. 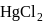 and
and 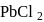 have same shape.
have same shape.
The statements which are not correct are
Detailed Solution: Question 8
The compound MX4 is tetrahedral. The number of ∠XMX formed in the compound are
Detailed Solution: Question 9
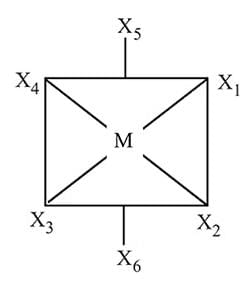
In a regular octahedral molecule, MX6 the number of X−M−X bonds at 180∘ is
Detailed Solution: Question 10
Which of the following molecular orbitals has two nodal planes?
Detailed Solution: Question 11
In the reaction 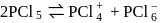 , the change in hybridisation is from
, the change in hybridisation is from
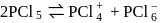 , the change in hybridisation is from
, the change in hybridisation is fromDetailed Solution: Question 12
Which of the following represents the given mode of hybridisation sp2 − sp2 − sp − sp from left to right ?
Detailed Solution: Question 13
Which of the following statement is incorrect?
(i) 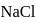 being an ionic compound is a good conductor of electricity in the solid state.
being an ionic compound is a good conductor of electricity in the solid state.
(ii) In canonical structures there is no difference in the arrangement of atoms.
(iii) Hybrid orbitals form stronger bonds than pure orbitals.
(iv) VSEPR theory can explain the square planar geometry of 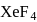 .
.
Detailed Solution: Question 14
Detailed Solution: Question 15
Detailed Solution: Question 16
In which one of the following pairs the two species have identical shape, but differ in hybridisation?
Detailed Solution: Question 17
Which one of the following molecules will form a linear polymeric structure due to H-bonding?
Detailed Solution: Question 18
On hybridization of one 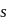 and one p-orbital we get
and one p-orbital we get
Detailed Solution: Question 19
Detailed Solution: Question 20
335 videos|699 docs|300 tests |


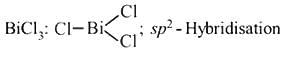

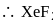 undergoes
undergoes 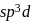 hybridisation, whereas
hybridisation, whereas 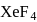 undergoes
undergoes 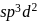 hybridisation.
hybridisation.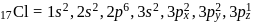
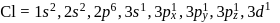
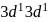
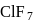 . Its shape is pentagonal bipyramidal.
. Its shape is pentagonal bipyramidal.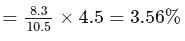
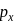 and
and 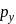 orbitals do not have proper orientation to overlap and hence no bond is formed.
orbitals do not have proper orientation to overlap and hence no bond is formed.
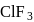 has 3 bond pairs and 2 lone pairs thus it is
has 3 bond pairs and 2 lone pairs thus it is 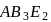 type molecule whereas
type molecule whereas 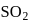 has
has 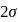 bond pairs and one lone pair so it is
bond pairs and one lone pair so it is 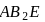 type molecule.
type molecule.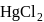 has a linear shape because it has 2 bond pairs only whereas
has a linear shape because it has 2 bond pairs only whereas  has 2 bond pairs with one lone pair thus it has a bent shape.
has 2 bond pairs with one lone pair thus it has a bent shape.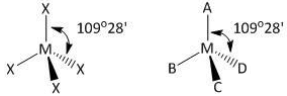
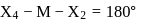
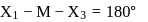
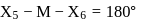
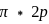
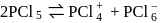
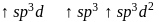

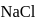 dissolves in water, the bond is broken and sodium and chlorine separate forming ions like
dissolves in water, the bond is broken and sodium and chlorine separate forming ions like 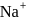 and
and 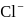 . Therefore, having free ions, it is able to conduct electricity. So, the option (1) is incorrect.
. Therefore, having free ions, it is able to conduct electricity. So, the option (1) is incorrect.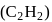
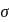 -bonds at
-bonds at 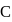 -atom
-atom 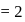
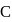 -atom
-atom 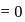
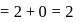
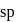 .
.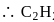 is linear
is linear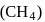
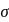 bonds at
bonds at 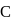 -atom
-atom 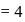
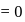
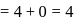
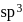 .
.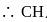 is tetrahedral.
is tetrahedral.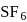 does not obey octet rule as in it S-atom has 12 electrons in its valence shell.
does not obey octet rule as in it S-atom has 12 electrons in its valence shell.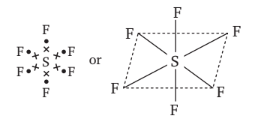
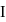 in
in 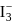 undergoes
undergoes 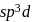 hybridisation giving a linear shape with three lone pairs at equatorial positions. Beryllium chloride has linear structure with
hybridisation giving a linear shape with three lone pairs at equatorial positions. Beryllium chloride has linear structure with 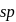 hybridisation of Be atom.
hybridisation of Be atom.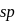 -hybrid orbitals is
-hybrid orbitals is 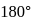 .
.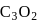 is
is