Ionic Bond and properties - Free MCQ Practice Test with solutions, JEE
MCQ Practice Test & Solutions: Test: Ionic Bond and properties (15 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Ionic Bond and properties". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which one of the following molecules contain both ionic and covalent bonds?
Detailed Solution: Question 1
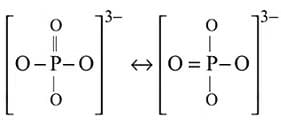
In 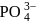 , the formal charge on each oxygen atom and the
, the formal charge on each oxygen atom and the  -
-  bond order respectively are
bond order respectively are
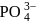 , the formal charge on each oxygen atom and the
, the formal charge on each oxygen atom and the 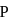 -
- 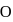 bond order respectively are
bond order respectively areDetailed Solution: Question 2
Which of the following is a favourable factor for cation formation?
Detailed Solution: Question 3
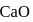 and
and 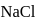 have the same crystal structure and approximately the same ionic radii. If
have the same crystal structure and approximately the same ionic radii. If 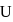 is the lattice energy of
is the lattice energy of 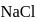 , the approximate lattice energy of
, the approximate lattice energy of 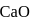 is
is
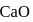 and
and 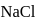 have the same crystal structure and approximately the same ionic radii. If
have the same crystal structure and approximately the same ionic radii. If 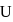 is the lattice energy of
is the lattice energy of 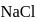 , the approximate lattice energy of
, the approximate lattice energy of 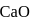 is
isDetailed Solution: Question 4
The compound which contains both ionic and covalent bonds is
Detailed Solution: Question 5
The electronegativity difference between 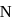 and
and 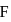 is greater than that between
is greater than that between 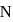 and
and 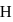 yet the dipole moment of
yet the dipole moment of 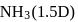 is larger than that of
is larger than that of 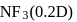 . This is because
. This is because
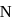 and
and 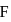 is greater than that between
is greater than that between 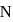 and
and 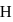 yet the dipole moment of
yet the dipole moment of 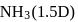 is larger than that of
is larger than that of 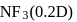 . This is because
. This is becauseDetailed Solution: Question 6
The dipole moments of diatomic molecules 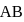 and
and 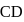 are
are 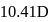 and
and 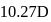 , respectively while their bond distances are
, respectively while their bond distances are 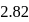 and
and 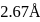 , respectively. This indicates that
, respectively. This indicates that
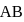 and
and 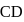 are
are 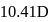 and
and 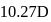 , respectively while their bond distances are
, respectively while their bond distances are 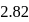 and
and 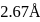 , respectively. This indicates that
, respectively. This indicates thatDetailed Solution: Question 7
If the bond length and dipole moment of a diatomic molecule are 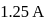 and
and 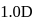 respectively, what is the per cent ionic character of the bond?
respectively, what is the per cent ionic character of the bond?
Detailed Solution: Question 8
The bond length of HCl molecule is 1.275Ã… and its dipole moment is 1.03D. The ionic character of the molecule (in percent) (charge of the electron = 4.8 × 10−10 esu) is
Detailed Solution: Question 9
What is the % ionic character of HCl if its observed dipole moment is 1.03D, electronic charge (q) is 4.8 × 10−10 e.s.u. and distance (d) between atoms is 1.27Å ?
Detailed Solution: Question 10
Which of the following statements is correct in the context of the allene molecule, 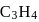 ?
?
Detailed Solution: Question 11
Detailed Solution: Question 12
The compound showing maximum covalent character is
Detailed Solution: Question 13
Amongst LiCl, RbCl, BeCl2 and MgCl2 the compounds with the greatest and the least ionic character, respectively are:
Detailed Solution: Question 14
The dipole moment of 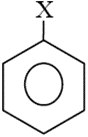 is
is 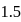
 . The dipole moment of
. The dipole moment of 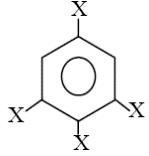 is
is
Detailed Solution: Question 15
335 videos|697 docs|300 tests |



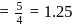
 atoms. Formal charge
atoms. Formal charge 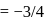
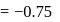

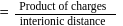
 the atomic dipole (orbital dipole due to lone pair) and bond dipole are in the same direction whereas in
the atomic dipole (orbital dipole due to lone pair) and bond dipole are in the same direction whereas in 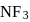 these are in opposite direction so in the former case they are added up whereas in the latter case net result is reduction of dipole moment. It has been shown in the following figure :
these are in opposite direction so in the former case they are added up whereas in the latter case net result is reduction of dipole moment. It has been shown in the following figure :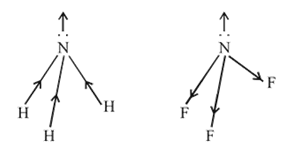
 electric charge
electric charge  bond length
bond length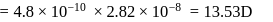
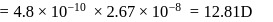
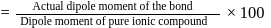 then
then 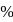 ionic character in
ionic character in 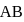
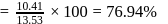
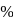 ionic character in
ionic character in 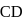
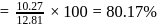
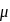 experimental
experimental 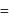 Dipole moment
Dipole moment 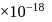
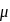 theoretical
theoretical 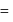 Bond length
Bond length 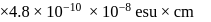
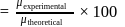
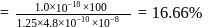



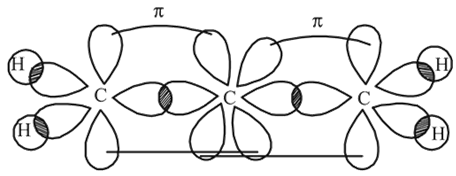
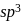 hybridisation forming four
hybridisation forming four 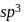 hybrid orbitals.
hybrid orbitals.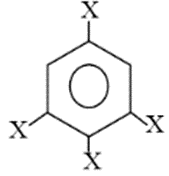
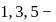 trihalobenzene is zero due to symmetry, thus its net dipole moment is same as halo benzene.
trihalobenzene is zero due to symmetry, thus its net dipole moment is same as halo benzene.