Ionic Equilibrium: Acid & Base - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Ionic Equilibrium: Acid & Base (12 Questions)
You can prepare effectively for NEET Topic-wise MCQ Tests for NEET with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Ionic Equilibrium: Acid & Base". These 12 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 15 minutes
- - Number of Questions: 12
Sign up on EduRev for free to attempt this test and track your preparation progress.
Direction (Q. Nos. 1-15) This section contains 15 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Q. Which of the following is least likely to behave as Lewis base?
Detailed Solution: Question 1
Which of the following is not acid-base conjugate pair?
Detailed Solution: Question 2
Which one of the following molecular hydrides acts as a Lewis acid?
Detailed Solution: Question 3
The species present in solution when CO2 dissolved in water, are
[IIT JEE 2006]
Detailed Solution: Question 4
In the following reaction,
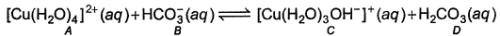
Species behaving as Bronsted-Lowry acids are
Species behaving as Bronsted-Lowry acids are
Detailed Solution: Question 5
Which of the following will produce a buffer solution when mixed in equal volumes ?
Detailed Solution: Question 6
Detailed Solution: Question 7
Detailed Solution: Question 8
Direction Choices for the correct combination of elements from Column I and Column II are given as options (a), (b), (c) and (d), out of which one is correct.
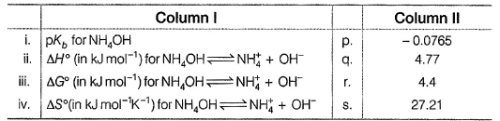
Q. The following thermodynamic quantities are given at 298 K.

Match the parameters given in Column I with their respective values given in Column II.
Codes
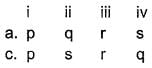
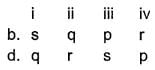
Match the parameters given in Column I with their respective values given in Column II.
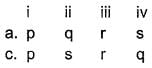
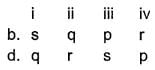
What will be the value of pH of 0.01 mol dm–3 CH3COOH (Ka = 1.74 × 10–5)?
Detailed Solution: Question 10
Detailed Solution: Question 11
For A- + H2O ⇔ HA + OH-, Kb = 1 x 10-12
Thus, pKa of HA + H2O ⇔ H3O+ + A- is ........
Detailed Solution: Question 12
6 docs|803 tests |