Isolation of Elements - 2 (Old NCERT) - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Isolation of Elements - 2 (Old NCERT) (30 Questions)
You can prepare effectively for JEE Chapter-wise Tests for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Isolation of Elements - 2 (Old NCERT)". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 45 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Formation of metallic copper from the sulphide ore in the commercial thermo-metallurgical process essentially involves which one of the following reaction :
Detailed Solution: Question 1
Ag2S + NaCN + Zn →Ag
This method of extraction of Ag by complex formation and then its displacement is called :
Detailed Solution: Question 2
Calcination is the process of heating the ore :
Detailed Solution: Question 3
Which of the following does not contain Mg :
Detailed Solution: Question 4
Match the method of concentration of the ore in column I with the ore in column II and select the correct alternate:
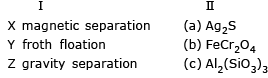
Detailed Solution: Question 5
Bessemerization is carried out for
(I) Fe
(II) Cu
(III) Al
(IV) silver
(II) Cu
(III) Al
(IV) silver
Detailed Solution: Question 6
Detailed Solution: Question 7
Which of the following is not used for obtaining Ag
Detailed Solution: Question 9
Which one of the following is not a method of concentration of ore?
Detailed Solution: Question 10
In which of the following isolations no reducing agent is required :
Detailed Solution: Question 11
Chemical leaching is useful in the concentration of :
Detailed Solution: Question 12
The element which could be extracted by electrolytic reduction of its oxide dissolved in a high temperature melt is :
Detailed Solution: Question 13
Consider the following statements :
Roasting is carried out to :
(i) convert sulphide to oxide and sulphate (ii) remove water of hydration
(iii) melt the ore (iv) remove arsenic and sulphur impurities
Of these statements :
Detailed Solution: Question 14
Iron obtained from blast furance is :
Detailed Solution: Question 15
Which one of the following statements is not correct :
Detailed Solution: Question 16
Which of the following is not an ore :
Detailed Solution: Question 17
In the extraction of nickel by Mond process, the metal is obtained by :
Detailed Solution: Question 18
Detailed Solution: Question 19
Froath floatation process is based on:
Detailed Solution: Question 20
When ZnS and PbS minerals are present together, then NaCN is added to separate them in the froth floatation process as a depressant, because
Detailed Solution: Question 21
When copper is purified by electrorefining process, noble metals like Ag and Au are found in
Detailed Solution: Question 22
Formation of Ni(CO)4 and subsequent its decomposition into Ni and CO (recycled) makes basis of Mond's process
Ni + 4CO 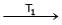 Ni (CO)4
Ni (CO)4 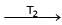 Ni + 4CO
Ni + 4CO
T1 and T2 are :
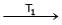 Ni (CO)4
Ni (CO)4 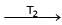 Ni + 4CO
Ni + 4CODetailed Solution: Question 23
Match column–I (process) with column–II (electrolyte) :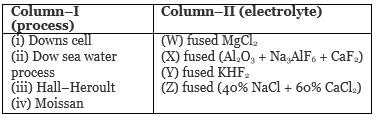
Choose the correct alternate.
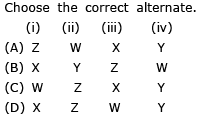

Detailed Solution: Question 24
(I) FeCr2O4 + NaOH + air → (A) + Fe2O3
(II) (A) + (B) → Na2Cr2O7
(III) Na2Cr2O7 + X 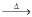 Cr2O3
Cr2O3
(IV) Cr2O3 + Y 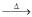 Cr
Cr
Q.
Compounds (A) and (B) are :
Detailed Solution: Question 25
(I) FeCr2O4 + NaOH + air → (A) + Fe2O3
(II) (A) + (B) → Na2Cr2O7
(III) Na2Cr2O7 + X 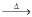 Cr2O3
Cr2O3
(IV) Cr2O3 + Y 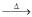 Cr
Cr
Q.
(X) and (Y) are :
Detailed Solution: Question 26
(I) FeCr2O4 + NaOH + air → (A) + Fe2O3
(II) (A) + (B) → Na2Cr2O7
(III) Na2Cr2O7 + X 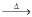 Cr2O3
Cr2O3
(IV) Cr2O3 + Y 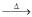 Cr
Cr
Q.
Na2CrO4 and Fe2O3 are separated by :
(I) FeCr2O4 + NaOH + air → (A) + Fe2O3
(II) (A) + (B) → Na2Cr2O7
(III) Na2Cr2O7 + X 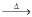 Cr2O3
Cr2O3
(IV) Cr2O3 + Y 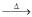 Cr
Cr
Q.
High temperature (> 1000ºC) electrolytic reduction is necessary for isolating :
Detailed Solution: Question 28
In froth-floatation process, palm oil functions as :
Detailed Solution: Question 29
Collector are the substances which help in attachement of an ore particle to air bubble in froth. A popular collector used industrially is :
Detailed Solution: Question 30
446 docs|929 tests |



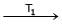 Ni (CO)4
Ni (CO)4 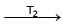 Ni + 4CO
Ni + 4CO