Isomerism of coordination compounds - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Isomerism of coordination compounds (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Isomerism of coordination compounds". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following molecule(s) is/are showing optical isomerism?
Detailed Solution: Question 1
The complex that exists as a pair of enantiomers is
Detailed Solution: Question 2
Which of the following pairs of structures represent facial and meridional isomers (geometrical isomers) respectively?
Detailed Solution: Question 3
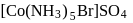 and
and 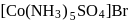 are example of which type of isomerism
are example of which type of isomerism
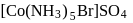 and
and 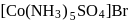 are example of which type of isomerism
are example of which type of isomerismDetailed Solution: Question 4
Which of the following types of isomerism is exhibited by
[Co(NH3)5(NO2)](NO3)2
i. Optical
ii. Linkage
iii. Ionization
iv. Coordination
[Co(NH3)5(NO2)](NO3)2
i. Optical
ii. Linkage
iii. Ionization
iv. Coordination
Detailed Solution: Question 5
Which among the following doesn't form a white precipitate with 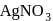 solution?
solution?
Detailed Solution: Question 6
Consider the following complexes.
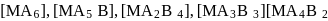
Find the number of complexes will show geometrical isomerism.
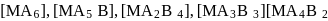
Find the number of complexes will show geometrical isomerism.
Detailed Solution: Question 7
Total number of geometrical isomers from the complex [RhCl(CO)(PPh3)(NH3)] is
Detailed Solution: Question 8
Which of the following complex will show geometrical as well as optical isomerism (en=ethylenediammine)
Detailed Solution: Question 9
[Co(NH3)5SO4]Br and [Co(NH3)5Br]SO4 are a pair of isomers.
Detailed Solution: Question 10
Which of the following will give maximum number of isomers?
Detailed Solution: Question 11
The complex given is
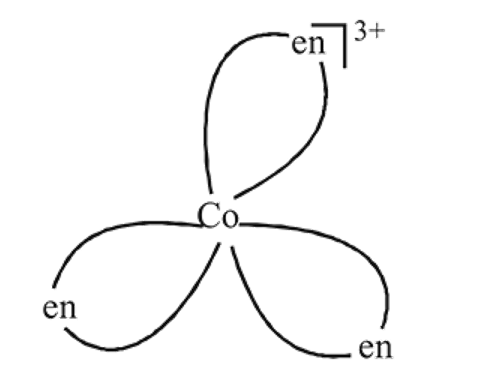
(i) non-superimposable on its mirror images
(ii) optically active
(iii) rotate plane polarised light
(iv) planar

(i) non-superimposable on its mirror images
(ii) optically active
(iii) rotate plane polarised light
(iv) planar
Detailed Solution: Question 12
Detailed Solution: Question 13
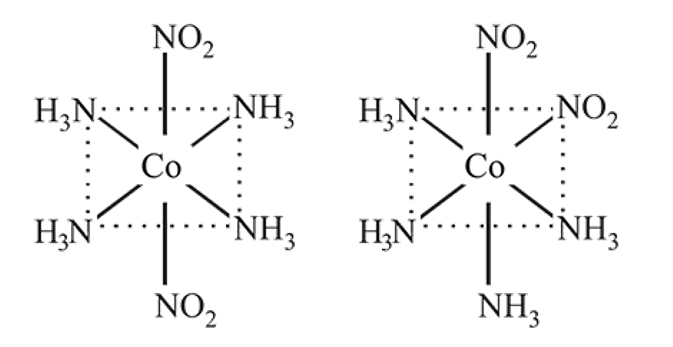
Coordination number of 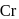 is six. A complex with
is six. A complex with 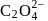 , en and superoxide
, en and superoxide 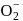 will be in the ratio to make complex
will be in the ratio to make complex 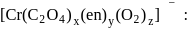
Detailed Solution: Question 14
For complex ion/compound formation reactions
(I) 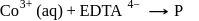
(II) 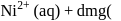 excess
excess 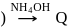
(III) 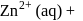 gly
gly 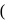 excess
excess 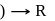
(IV) 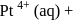 en
en 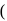 excess
excess 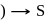
which of the following complex ion/compound does not exhibit optical activity?
(I)
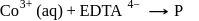
(II)
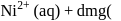 excess
excess 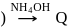
(III)
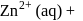 gly
gly 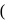 excess
excess 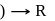
(IV)
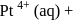 en
en 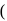 excess
excess 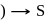
which of the following complex ion/compound does not exhibit optical activity?
Detailed Solution: Question 15
An octahedral complex with molecular composition 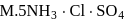 has two isomers,
has two isomers,
A and 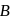 . The solution of
. The solution of 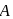 gives a white precipitate with
gives a white precipitate with 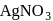 solution and the solution of B gives white precipitate with
solution and the solution of B gives white precipitate with 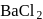 solution. The type of isomerism exhibited by the complex is:
solution. The type of isomerism exhibited by the complex is:
Detailed Solution: Question 16
Type of isomerism which exists between 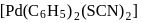 and
and 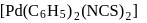 is :
is :
Detailed Solution: Question 17
Which of the following complex ions will exhibit optical isomerism? 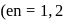 -diamine ethane
-diamine ethane  .
.
Detailed Solution: Question 18
Detailed Solution: Question 19
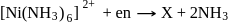
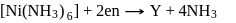
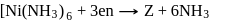
Which of them show optical as well as geometrical isomerism?
Detailed Solution: Question 20
335 videos|699 docs|300 tests |


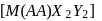 containing one symmetrical bidentate ligand.
containing one symmetrical bidentate ligand.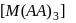
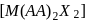
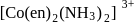

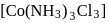 exhibits fac-mer isomerism.
exhibits fac-mer isomerism.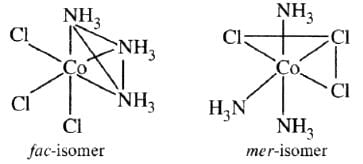
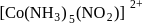 and
and 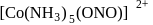
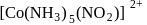 and
and 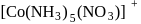
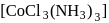 complex contains elements of symmetry and is optically inactive. It does not give white precipitate with silver nitrate solution as chlorine is present in coordination sphere.
complex contains elements of symmetry and is optically inactive. It does not give white precipitate with silver nitrate solution as chlorine is present in coordination sphere.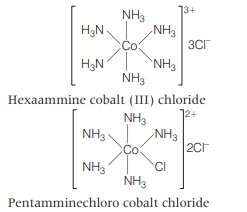
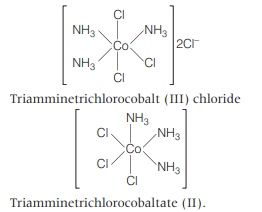
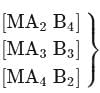 Each show 2 geometical isomers
Each show 2 geometical isomers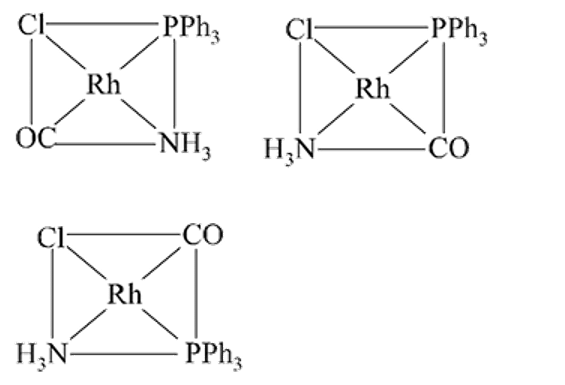
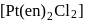 is a complex of the type
is a complex of the type 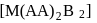 which is octahedral. Such compounds exhibit optical and geometrical isomerism both.
which is octahedral. Such compounds exhibit optical and geometrical isomerism both.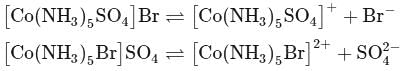
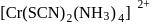 can
can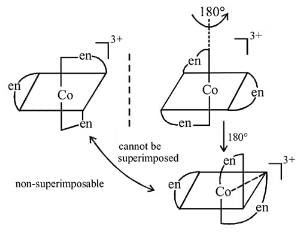
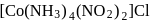 exhibits
exhibits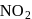 group which may be in the form
group which may be in the form 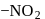 or -ONO.
or -ONO. 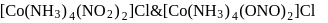
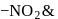
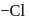
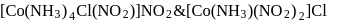
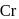 is 6
is 6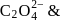 en are bidentate ligands while
en are bidentate ligands while 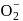 is unidentate
is unidentate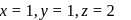
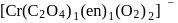
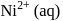 with DMG (dimethylglyoxime) form square planar complex which is obtically inactive. While
with DMG (dimethylglyoxime) form square planar complex which is obtically inactive. While 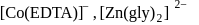 and
and 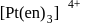 are optically active as they do not have P.O.S.
are optically active as they do not have P.O.S.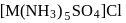 and
and 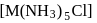
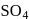 . They respectively give chloride ion (indicated by precipitation with
. They respectively give chloride ion (indicated by precipitation with 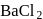 ) and
) and 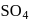 ion (indicated by precipitation with
ion (indicated by precipitation with 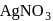 ). Hence the type of isomerism exhibited by the complex is ionization isomerism.
). Hence the type of isomerism exhibited by the complex is ionization isomerism.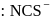 and
and 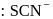
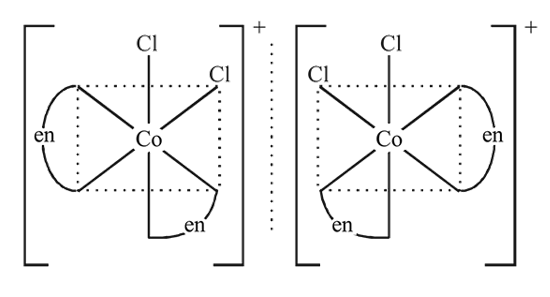
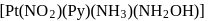
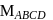 may exist in three isomeric form. Similarily
may exist in three isomeric form. Similarily 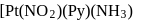
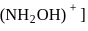 may exist in three isomeric form.
may exist in three isomeric form.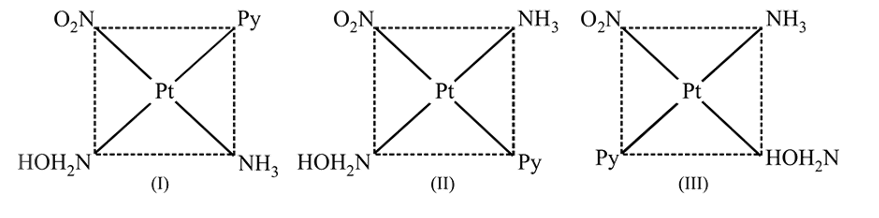
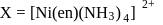 neither G.I. nor OI
neither G.I. nor OI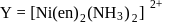 G. I as well as
G. I as well as 
 does not show G.I. while
does not show G.I. while