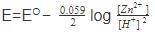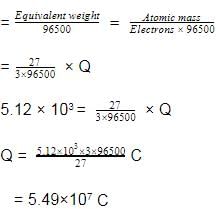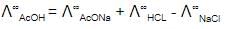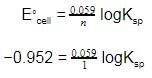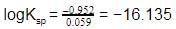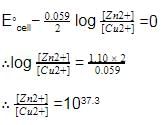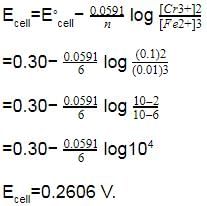JEE Previous Year Questions- Electrochemistry with solutions - Free MCQ
MCQ Practice Test & Solutions: Test: JEE Previous Year Questions- Electrochemistry (21 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: JEE Previous Year Questions- Electrochemistry". These 21 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 30 minutes
- - Number of Questions: 21
Sign up on EduRev for free to attempt this test and track your preparation progress.
For the following cell with hydrogen electrodes at two different pressure p1 and p2
Pt(H2)|H+ (aq.)|Pt(H2)| H+ (aq.) emf is given by – p1 p2
[AIEEE-2002]
Which of the following reactions is possible at anode ?
[AIEEE-2002]
Detailed Solution: Question 2
For a cell given below Ag | Ag+ || Cu2+ | Cu
— +
Ag+ + e- → Ag, Eº = x
Cu2+ +2e- → Cu, Eº = y
Eº cell is –
[AIEEE-2002]
Detailed Solution: Question 3
For a cell reaction involving a two-electron change the standard e.m.f. of the cell is found to be 0.295 V at 25ºC. The equilibrium constant of the reaction at 25º C will be –
[AIEEE-2003]
Detailed Solution: Question 4
Standard reduction electrode potentials of three metals A, B and C are respectively +0.5 V, – 3.0V and – 1.2 V. The reducing powers of these metals are -
[AIEEE-2003]
Detailed Solution: Question 5
For the redox reaction :
Zn (s) + Cu2+ (0.1M) → Zn2+ (1M) + Cu(s) taking place in a cell, Eºcell is 1.10 volt. Ecell for the cell will be
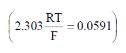
[AIEEE-2003]
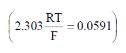
Detailed Solution: Question 6
When during electrolysis of a solution of AgNO3 9650 coulombs of charge pass through the electroplating bath, the mass of silver deposited on the cathode will be –
[AIEEE-2003]
Detailed Solution: Question 7
Consider the following E° values
[AIEEE-2004]
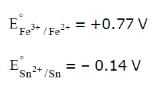
under standard conditions the potential for the reaction
Sn(s) + 2Fe3+ (aq) → 2Fe2+ (aq) + Sn2+ (aq) is
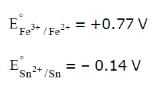
Detailed Solution: Question 8
The standard e.m.f. of a cell, involving one electron change is found to be 0.591 V at 25°C. The equilibrium constant of the reaction is (F = 96500 C mol-1 ; R = 8.314 JK-1mol-1)
[AIEEE-2004]
Detailed Solution: Question 9
The limiting molar conductivities ^° for NaCl, KBr and KCl are 126, 152 and 150 S cm2 mol-1 respectively. The ^° for NaBr is –
[AIEEE-2004]
Detailed Solution: Question 10
In a cell that utilises the reaction
Zn(s) + 2H+ (aq)  Zn2+ (aq) + H2(g)
Zn2+ (aq) + H2(g)
addition of H2SO4 to cathode compartment, will–
[AIEEE-2004]
Detailed Solution: Question 11
Aluminium oxide may be electrolysed at 1000ºC to furnish aluminium metal (At. Mass=27 amu ; 1 Faraday = 96,500 Coulombs). The cathode reaction is
Al3+ + 3e-→ Alº
To prepare 5.12 kg of aluminium metal by this method would require -
[AIEEE-2005]
Detailed Solution: Question 12

Calculate  using appropriate molar conductance of the electrolytes listed above at infinite dilution in H2O at 25ºC
using appropriate molar conductance of the electrolytes listed above at infinite dilution in H2O at 25ºC
[AIEEE-2005]

Detailed Solution: Question 13
For a spontaneous reaction the ΔG, equilibrium constant (K) and Ecell will be respectively -
[AIEEE-2005]
Detailed Solution: Question 14
Given the data at 25ºC,
Ag + I- → AgI + e- Eº = 0.152 V
Ag → Ag+ + e- Eº = -0.800 V
What is the value of log Ksp for AgI ?
(2.303 RT/F = 0.059 V)
[AIEEE 2006]
Detailed Solution: Question 15
The cell, Zn | Zn2+ (1M) | | Cu2+(1M)|Cu (Eºcell)=1.10 V), was allowed to be completely discharged at 298 K.
the relative concentration of zn2+ to 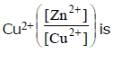
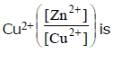
Detailed Solution: Question 16
The equivalent conductance of two strong electrolytes at infinite dilution in H2O (where ions move freely through a solution) at 25ºC are given below -
^°CH3COONa= 91.0 S cm2/equiv
^ºHCl = 426.2 S cm2/equiv
What additional information/quantity one needs to calculate Lº of an aqueous solution of acetic acid ?
[AIEEE 2007]
Given EºCr3+/Cr = – 0.72 V, EºFe2+/Fe= – 0.42 V. The potential for the cell Cr |Cr3+ (0.1 M)| |Fe2+ (0.01 M) | Fe is -
[AIEEE 2008]
Detailed Solution: Question 18
The standard reduction potentials for Zn2+/Zn, Ni2+ /Ni, and Fe2+/Fe are – 0.76, – 0.23 and – 0.44 V respectively. The reaction X + Y2+ → X2+ + Y will be spontaneous when –
[AIEEE 2012]
Detailed Solution: Question 19
Given
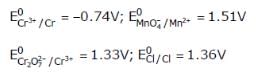
Based on the data given above, strongest oxidisting agent will be
[AIEEE 2013]
Detailed Solution: Question 20
Refining of impure copper with zinc impurity is to be done by electrolysis using electrods as –
[AIEEE-2002]
Detailed Solution: Question 21
335 videos|699 docs|300 tests |