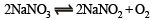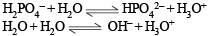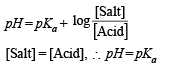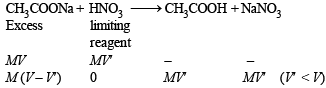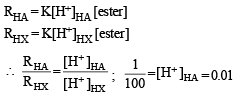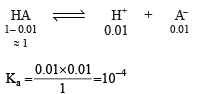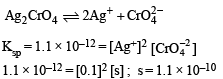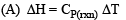MCQs (One or More Correct Option): Equilibrium | JEE Advanced - Free MCQ
MCQ Practice Test & Solutions: Test: MCQs (One or More Correct Option): Equilibrium | JEE Advanced (12 Questions)
You can prepare effectively for JEE Crack JEE with 35 Years of Previous Year Solved Papers with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: MCQs (One or More Correct Option): Equilibrium | JEE Advanced". These 12 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 12
Sign up on EduRev for free to attempt this test and track your preparation progress.
For the gas phase reaction :
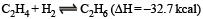
carried out in a vessel, the equilibrium concentration of C2H4 can be increased by :
Detailed Solution: Question 1
When NaNO3 is heated in a closed vessel, oxygen is liberated and NaNO2 is left behind. At equilibrium.
Detailed Solution: Question 2
The equilibrium :

is attained at 25ºC in a closed container and an inert gas, helium is introduced. Which of the following statements are correct?

Detailed Solution: Question 3
For the reaction :
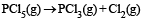
The forward reaction at constant temperature is favoured by
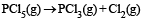
Detailed Solution: Question 4
For the reaction 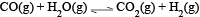 at a given temperature the equilibrium amount of CO2(g) can be increased by
at a given temperature the equilibrium amount of CO2(g) can be increased by
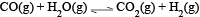 at a given temperature the equilibrium amount of CO2(g) can be increased by
at a given temperature the equilibrium amount of CO2(g) can be increased byDetailed Solution: Question 5
Which of the following statements(s) is (are) correct?
Detailed Solution: Question 6
A buffer solution can be prepared from a mixture of
Detailed Solution: Question 7
Aqueous solutions of HNO3 , KOH, CH3COOH and CH3COONa of identical concentrations are provided. The pair(s) of solutions which form a buffer upon mixing is(are)
Detailed Solution: Question 8
The initial rate of hydrolysis of methyl acetate (1M) by a weak acid (HA, 1M) is 1/100th of that of a strong acid (HX, 1M), at 25°C. The Ka of HA is
Detailed Solution: Question 9
The Ksp of Ag2CrO4 is 1.1 × 10–12 at 298 K. The solubility (in mol/L) of Ag2CrO4 in a 0.1 M AgNO3 solution is
Detailed Solution: Question 10
The thermal dissociation equilibrium of CaCO3(s) is studied under different conditions
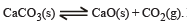
For this equilibrium, the correct statement(s) is(are)
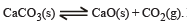
Detailed Solution: Question 11
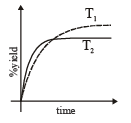
The %yield of ammonia as a function of time in the reaction is given below
is given below
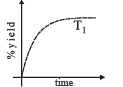
If this reaction is conducted at (P, T2), with T2 > T1, the %yield of ammonia as a function of time is represented by
 is given below
is given below
Detailed Solution: Question 12
347 docs|185 tests |