Noble gases and its properties - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Noble gases and its properties (15 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Noble gases and its properties". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
Compounds formed when the noble gases get entrapped in the cavities of crystal lattices of certain organic and inorganic compounds are known as
Detailed Solution: Question 1
Ionisation potential values of noble gases decrease down the group with increase in atomic size. Xenon forms binary fluorides by the direct reaction of elements. Identify the correct statement(s) from below.
Detailed Solution: Question 2
Detailed Solution: Question 3
Incorrect statement regarding following reactions is:
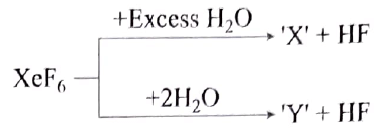
Detailed Solution: Question 4
MF + XeF4 ⟶ ′A′(M+ = Alkali metal cation)
The state of hybridisation of the central atomin 'A' and shape of the species are:
The state of hybridisation of the central atomin 'A' and shape of the species are:
Detailed Solution: Question 5
Detailed Solution: Question 6
Detailed Solution: Question 7
When helium gas is allowed to expand into vaccum, heating effect is observed. The reason for this is (assume He as a non ideal gas)
Detailed Solution: Question 8
Detailed Solution: Question 9
The ionization energy of 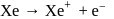 is very close to that of
is very close to that of
Detailed Solution: Question 10
When a mixture of 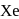 and
and 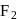 in the ratio of
in the ratio of 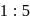 is heated at
is heated at 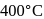 in a sealed nickel vessel, the compound formed is
in a sealed nickel vessel, the compound formed is
Detailed Solution: Question 11
Detailed Solution: Question 12
Detailed Solution: Question 13
Which of the following statements is incorrect?
Detailed Solution: Question 14
Detailed Solution: Question 15
332 videos|699 docs|300 tests |



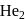 :
: 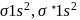
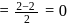
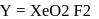 . No, it is not an oxyacid of
. No, it is not an oxyacid of 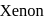
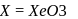 . Yes, it is explosive
. Yes, it is explosive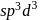 , pentagonal planar
, pentagonal planar . Tos atomic weight is
. Tos atomic weight is 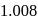 . But among the mentioned gases
. But among the mentioned gases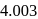 .
.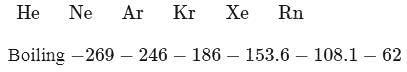
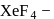 This compound is formed by xenon.
This compound is formed by xenon.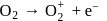
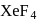
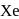 in
in 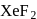 is
is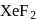 has
has
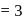
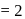
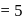
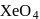 is
is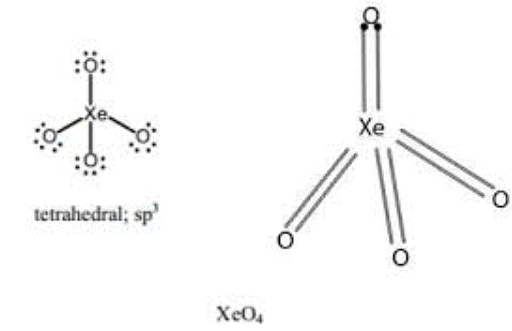
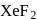 molecule has a trigonal bipyramidal structure.
molecule has a trigonal bipyramidal structure.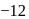 is
is